CD163-Positive Macrophage Enrichment in Peritoneal Implants of Serous Ovarian Tumors: A Quantitative Digital Pathology Study
Downloads
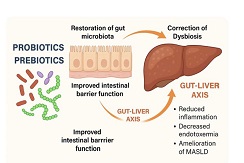
Peritoneal dissemination is a defining feature of epithelial ovarian cancer and plays a central role in disease progression and poor clinical outcome. Increasing evidence suggests that the tumor microenvironment, particularly tumor-associated macrophages, contributes significantly to metastatic behavior. CD163 is a well-established marker of M2-polarized macrophages associated with immune suppression and tumor promotion. However, the spatial distribution and quantitative characteristics of CD163-positive macrophages in peritoneal implants of serous ovarian tumors remain insufficiently characterized.
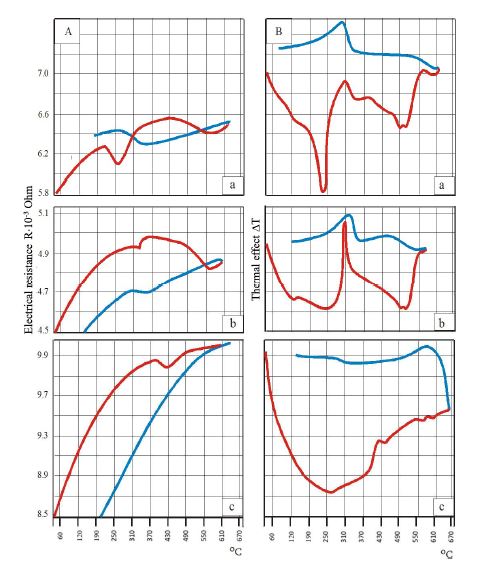
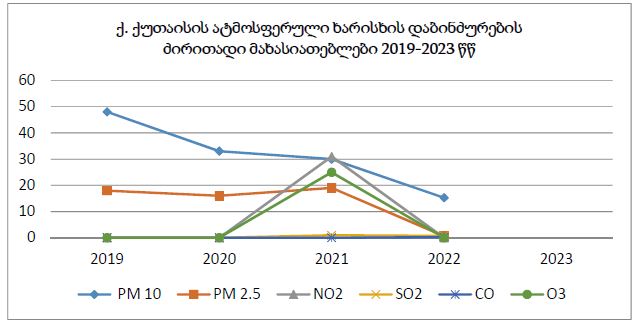
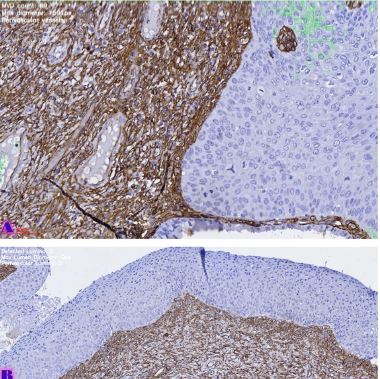
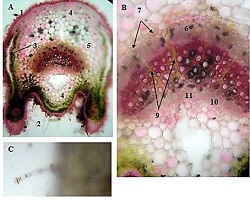
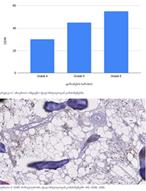
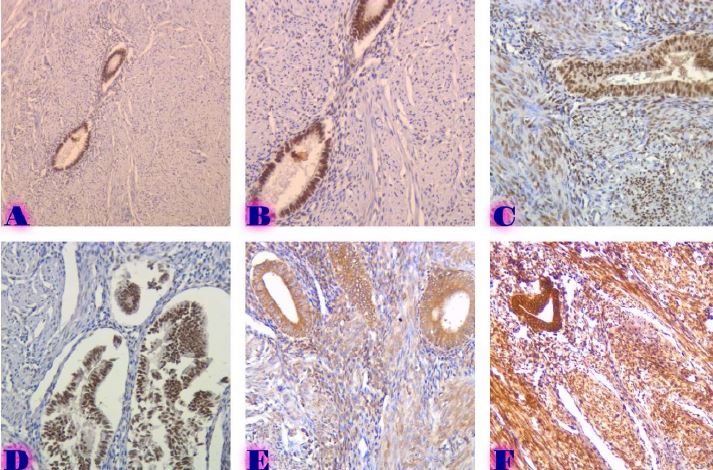
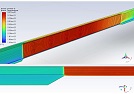
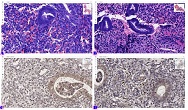
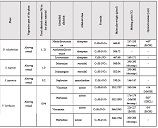
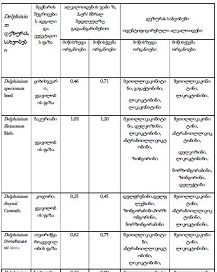
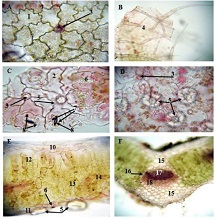
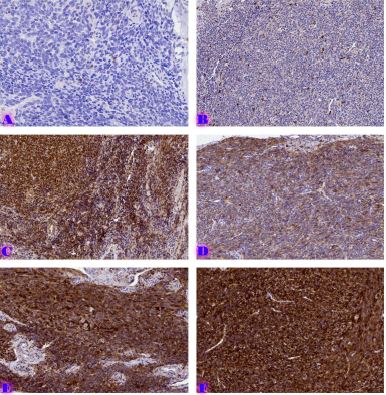
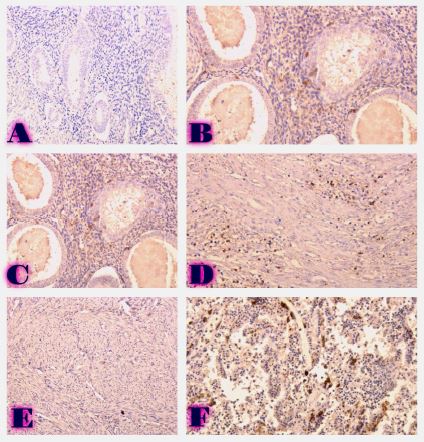
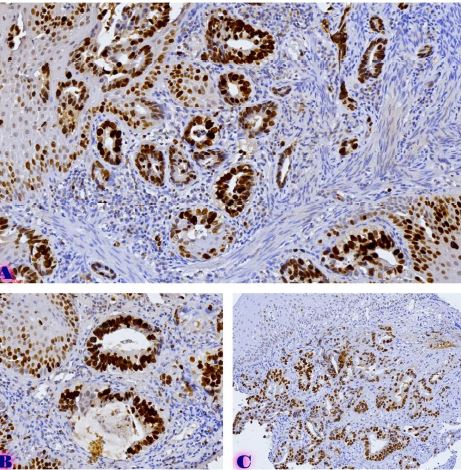
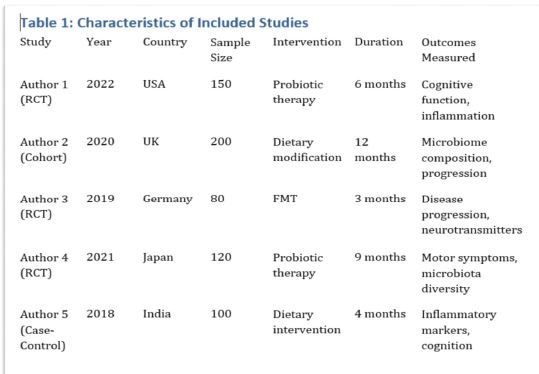
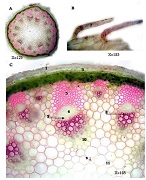
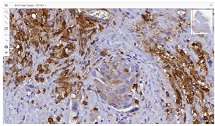
This study aimed to evaluate CD163-positive macrophage infiltration across the spectrum of serous ovarian tumors and to analyze their spatial distribution within peritoneal implants. A total of 42 cases, including serous borderline tumors, low-grade serous carcinoma, and high-grade serous carcinoma, were retrospectively analyzed. Immunohistochemical staining for CD163 was performed on formalin-fixed, paraffin-embedded tissue sections. Digital whole-slide imaging and quantitative analysis were used to assess macrophage density (cells/mm²) in predefined compartments, including stromal areas, tumor center, invasive front, and tumor–stroma interface.
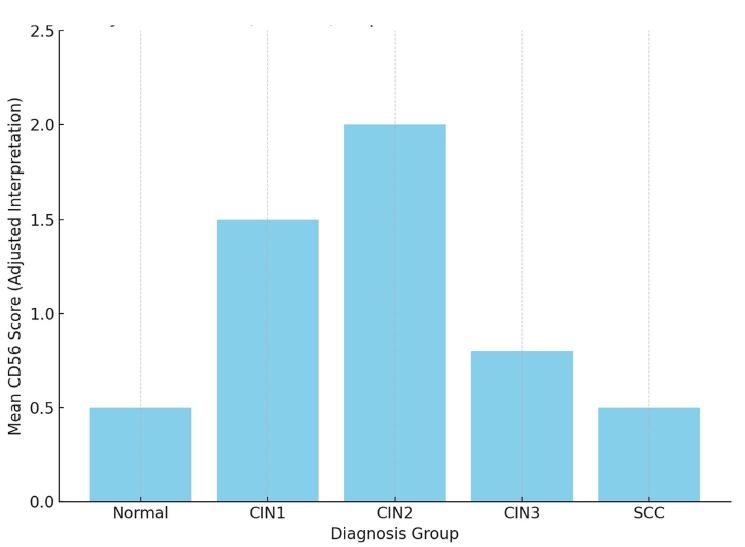
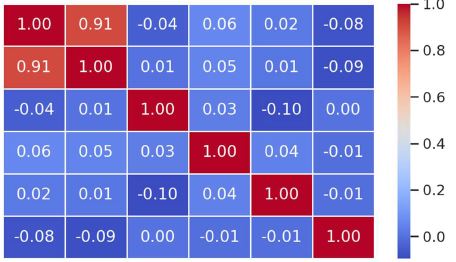
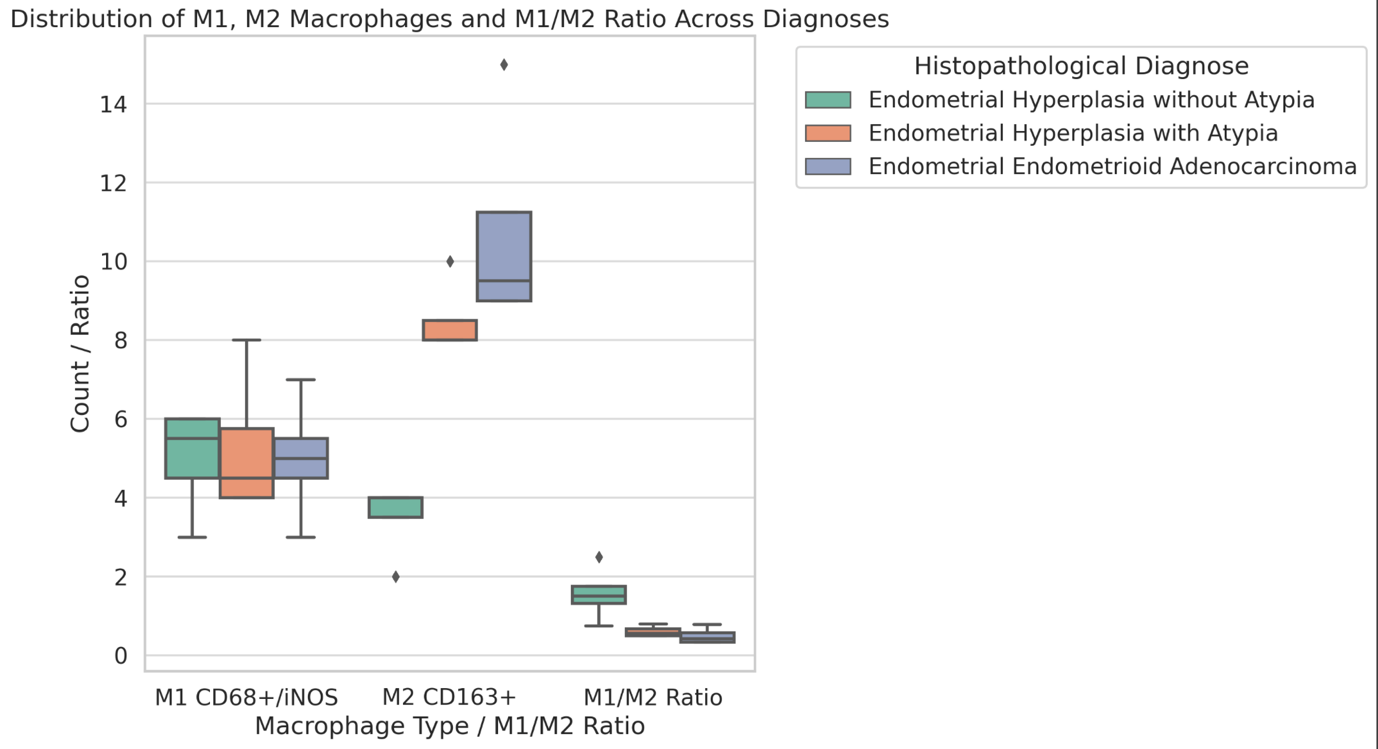
A statistically significant increase in CD163-positive macrophage density was observed across the histological spectrum, with the lowest values in borderline tumors and the highest in high-grade serous carcinoma. The most pronounced differences were identified in the stromal compartment, invasive front, and tumor–stroma interface (p < 0.001). Spatial analysis demonstrated preferential accumulation of macrophages in peritumoral regions associated with invasion, while tumor center regions showed comparatively lower densities. Strong correlations were identified between macrophage densities in stromal and invasive compartments, indicating coordinated microenvironmental remodeling.
These findings demonstrate progressive enrichment and spatial redistribution of CD163-positive macrophages in peritoneal implants of serous ovarian tumors. The observed patterns support the role of macrophage-mediated immune modulation and stromal interaction in tumor progression and metastatic dissemination. Quantitative spatial assessment of CD163-positive macrophages may provide valuable insight into tumor biology and represents a potential approach for microenvironment-based stratification in ovarian cancer.
Downloads
Devadze R, Gvenetadze A, Burkadze G, Kepuladze S. Distribution of tumor-associated macrophages and M1/M2 polarization in different types and grades of ovarian tumors. Indian Journal of Pathology and Oncology Journal homepage: www.ijpo.co.in Original Research Article [homepage on the Internet] 2022 [cited 2026 Apr 30];9(4):318–321. Available from: https://doi.org/10.18231/j.ijpo.2022.076
Heidarpour M, Hoseini-Beheshti M-S, Id M, Derakhshan ID. Evaluation of the relationship between CD163 positive macrophages and prognostic factors in serous ovarian tumors. Immunopathol Persa [homepage on the Internet] 2020 [cited 2026 Apr 30];6(2):23. Available from: www.immunopathol.comm
Hagemann T, Wilson J, Burke F, et al. Ovarian Cancer Cells Polarize Macrophages Toward A Tumor-Associated Phenotype. The Journal of Immunology 2006;176(8):5023–5032.
Duluc D, Corvaisier M, Blanchard S, et al. Interferon-γ reverses the immunosuppressive and protumoral properties and prevents the generation of human tumor-associated macrophages. Int J Cancer 2009;125(2):367–373.
Alvero AB, Montagna MK, Craveiro V, Liu L, Mor G. Distinct Subpopulations of Epithelial Ovarian Cancer Cells Can Differentially Induce Macrophages and T Regulatory Cells Toward a Pro-Tumor Phenotype. American Journal of Reproductive Immunology 2012;67(3):256–265.
Alvero AB, Chen R, Fu HH, et al. Molecular phenotyping of human ovarian cancer stem cells unravel the mechanisms for repair and chemo-resistance. Cell Cycle 2009;8(1):158–166.
Biswas SK, Gangi L, Paul S, et al. A distinct and unique transcriptional program expressed by tumor-associated macrophages (defective NF-κB and enhanced IRF-3/STAT1 activation). Blood 2006;107(5):2112–2122.
Schutyser E, Struyf S, Proost P, et al. Identification of biologically active chemokine isoforms from ascitic fluid and elevated levels of CCL18/pulmonary and activation-regulated chemokine in ovarian carcinoma. Journal of Biological Chemistry 2002;277(27):24584–24593.
Kryczek I, Zou L, Rodriguez P, et al. B7-H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. Journal of Experimental Medicine 2006;203(4):871–881.
No JH, Moon JM, Kim K, Kim YB. Prognostic significance of serum soluble CD163 level in patients with epithelial ovarian cancer. Gynecol Obstet Invest 2013;75(4):263–267.
Chen R, Alvero AB, Silasi DA, Mor G. Inflammation, cancer and chemoresistance: Taking advantage of the toll-like receptor signaling pathway. American Journal of Reproductive Immunology 2007;57(2):93–107.
Kulbe H, Chakravarty P, Leinster DA, et al. A dynamic inflammatory cytokine network in the human ovarian cancer microenvironment. Cancer Res 2012;72(1):66–75.
Reinartz S, Schumann T, Finkernagel F, et al. Mixed-polarization phenotype of ascites-associated macrophages in human ovarian carcinoma: Correlation of CD163 expression, cytokine levels and early relapse. Int J Cancer 2014;134(1):32–42.
Torroella-Kouri M, Silvera R, Rodriguez D, et al. Identification of a subpopulation of macrophages in mammary tumor-bearing mice that are neither M1 nor M2 and are less differentiated. Cancer Res 2009;69(11):4800–4809.
Zhang M, He Y, Sun X, et al. A high M1/M2 ratio of tumor-associated macrophages is associated with extended survival in ovarian cancer patients. J Ovarian Res 2014;7(1).
Robinson-Smith TM, Isaacsohn I, Mercer CA, et al. Macrophages mediate inflammation-enhanced metastasis of ovarian tumors in mice. Cancer Res 2007;67(12):5708–5716.
Neyen C, Plüddemann A, Mukhopadhyay S, et al. Macrophage Scavenger Receptor A Promotes Tumor Progression in Murine Models of Ovarian and Pancreatic Cancer. The Journal of Immunology 2013;190(7):3798–3805.
Tomšová M, Melichar B, Sedláková I, Šteiner I. Prognostic significance of CD3+ tumor-infiltrating lymphocytes in ovarian carcinoma. Gynecol Oncol 2008;108(2):415–420.
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: Tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 2002;23(11):549–555.
Zhorzholiani P, Bokhua Z, Kepuladze S, Burkadze G. The Role of M1 and M2 Macrophages in the Progression of Endometrial Hyperplasia to Endometrioid Adenocarcinoma. Georgian Scientists [homepage on the Internet] 2025 [cited 2026 Apr 30];7(1):24–37. Available from: https://journals.4science.ge/index.php/GS/article/view/3371
Takaishi K, Komohara Y, Tashiro H, et al. Involvement of M2-polarized macrophages in the ascites from advanced epithelial ovarian carcinoma in tumor progression via Stat3 activation. Cancer Sci 2010;101(10):2128–2136.
Kawamura K, Komohara Y, Takaishi K, Katabuchi H, Takeya M. Detection of M2 macrophages and colony-stimulating factor 1expression in serous and mucinous ovarian epithelial tumors. Pathol Int 2009;59(5):300–305.
Copyright (c) 2026 Georgian Scientists

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.