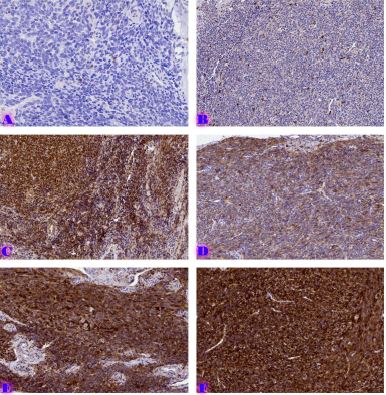
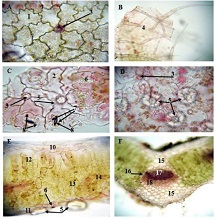
Expression patterns of hormone receptors and proliferative-apoptotic markers in cases of uterine adenomyosis, endometriosis, and endometriosis-associated carcinoma: a retrospective immunohistochemical study
Downloads
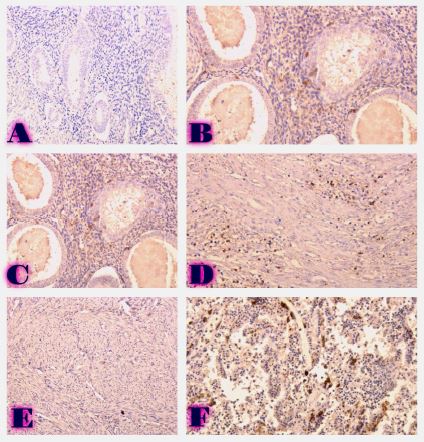
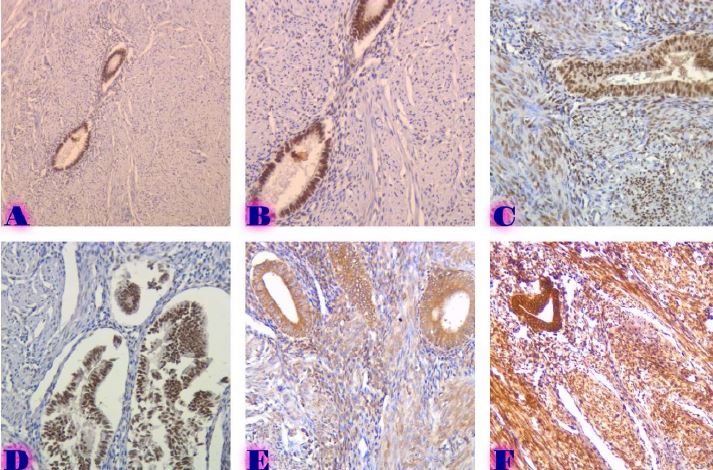
Background: Adenomyosis and endometriosis are estrogen-dependent disorders characterized by ectopic endometrial proliferation. While traditionally considered benign, atypical endometriosis exhibits histological and molecular features associated with neoplastic transformation, particularly in the development of endometriosis-associated carcinoma. This study evaluates the immunohistochemical expression of ER, PR, Ki67, BCL2, and P53 in eutopic endometrium, adenomyosis, endometriosis, atypical endometriosis, and endometriosis-associated carcinoma to assess their role in disease progression.
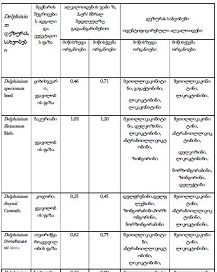
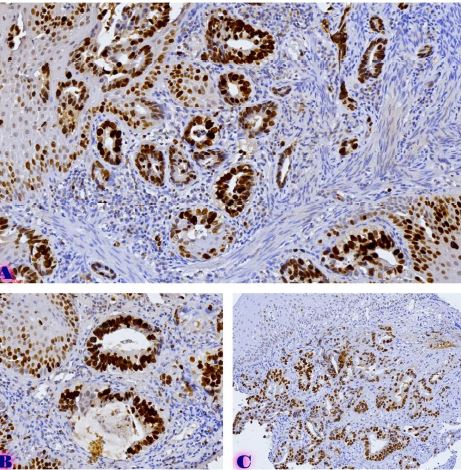
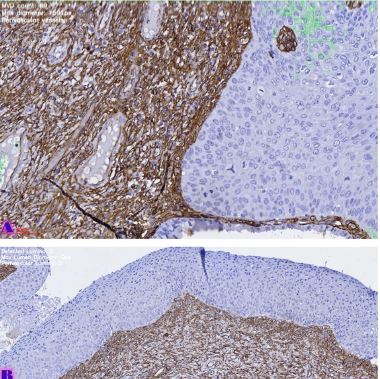
Methods: A total of 77 cases were retrospectively analyzed, including 21 adenomyosis, 17 eutopic endometrium, 14 endometriosis, 9 atypical endometriosis, and 16 endometriosis-associated carcinoma cases. Immunohistochemistry (IHC) was performed using Leica Bond-III automated staining system with ER (SP1), PR (16), Ki67 (MIB-1), BCL2 (124), and P53 (DO-7) antibodies. Expression levels were quantitatively assessed, and statistical analyses were conducted to compare marker expression across groups.
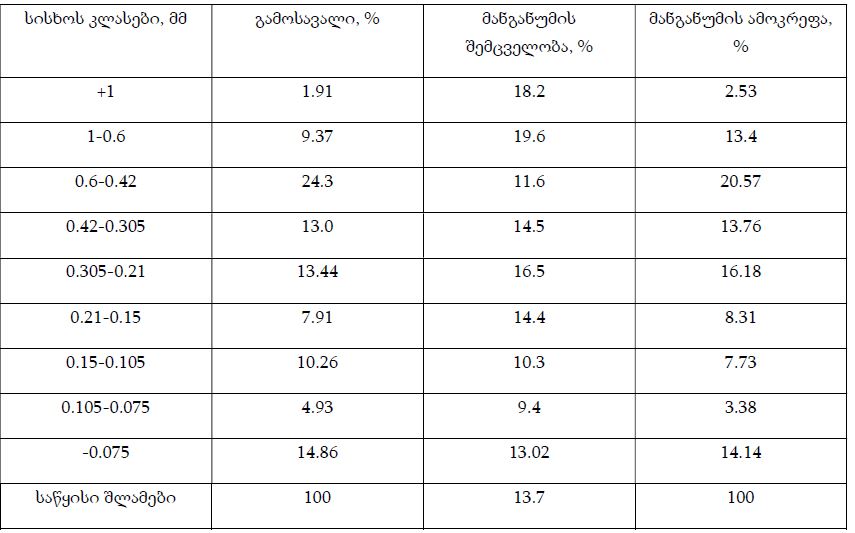
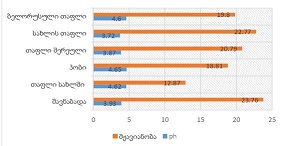
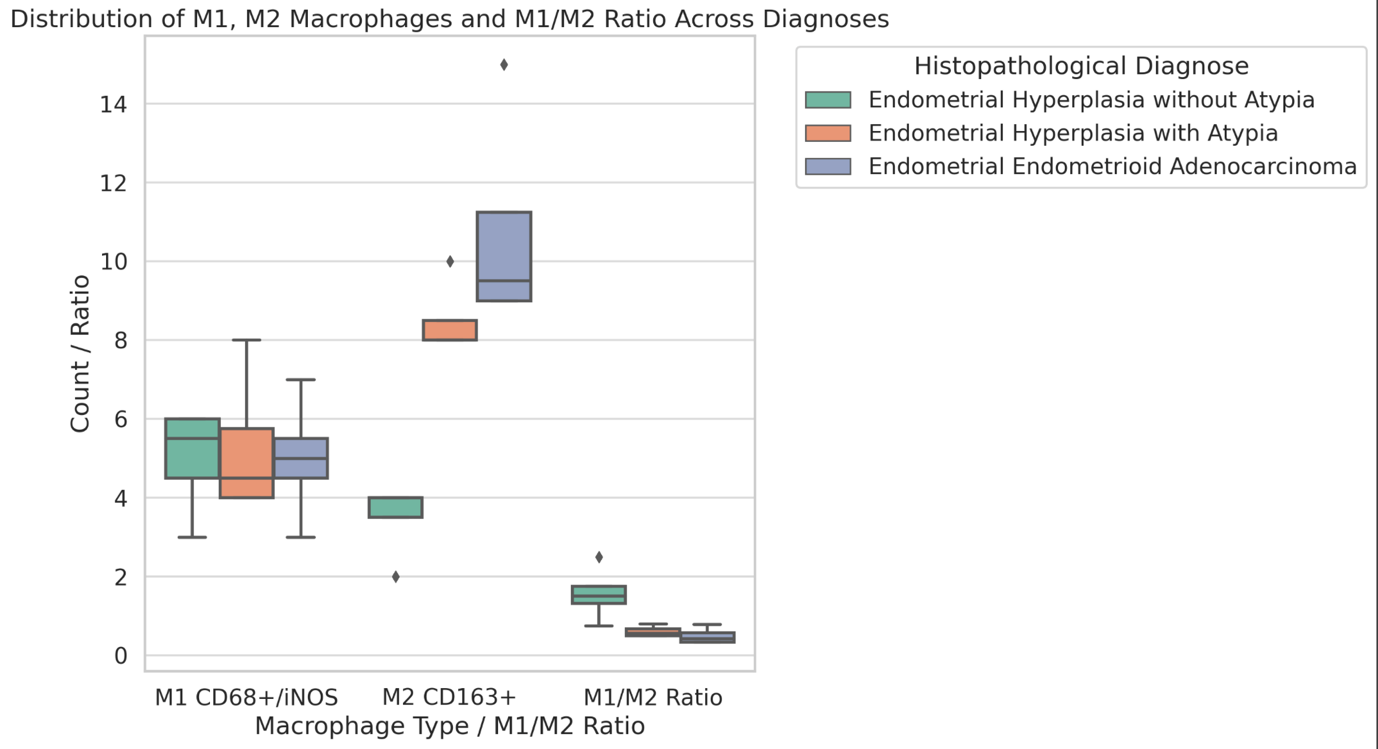
Results: ER and PR expression decreased significantly from eutopic endometrium to carcinoma (p < 0.0001), with atypical endometriosis exhibiting lower receptor expression than typical endometriosis (p = 0.002); Ki67 was significantly elevated in atypical endometriosis (p = 0.004) and highest in carcinoma (p < 0.0001), suggesting increased proliferative activity during progression; BCL2 expression was significantly lower in atypical endometriosis and carcinoma (p < 0.0001), indicating a shift from apoptotic resistance to dysregulation; P53 overexpression was observed in atypical endometriosis (p = 0.002) and further increased in carcinoma (p < 0.0001), supporting its role in malignant transformation.
Conclusion: Progressive hormonal receptor loss, increased proliferation, and apoptotic dysregulation were observed in atypical endometriosis and carcinoma, supporting the hypothesis that atypical endometriosis represents an intermediate stage toward malignancy. Immunohistochemical profiling of ER, PR, Ki67, BCL2, and P53 may aid in risk stratification and early identification of high-risk lesions. Further studies incorporating molecular and genomic analyses are warranted to refine the understanding of endometriosis-associated malignancies.
Downloads
Y. Cao et al., “Adenomyosis associated infertility: an update on immunological perspective,” Reprod Biomed Online, vol. 0, no. 0, p. 104703, Nov. 2024, doi: 10.1016/J.RBMO.2024.104703.
P. R. Koninckx, C. Meuleman, S. Demeyere, E. Lesaffre, and F. J. Cornillie, “Suggestive evidence that pelvic endometriosis is a progressive disease, whereas deeply infiltrating endometriosis is associated with pelvic pain.,” Fertil Steril, vol. 55, no. 4, pp. 759–765, 1991, doi: 10.1016/S0015-0282(16)54244-7.
J. Ottolina et al., “Endometriosis and Adenomyosis: Modern Concepts of Their Clinical Outcomes, Treatment, and Management,” J Clin Med, vol. 13, no. 14, p. 3996, Jul. 2024, doi: 10.3390/JCM13143996.
H. S. Taylor et al., “Treatment of Endometriosis-Associated Pain with Elagolix, an Oral GnRH Antagonist,” New England Journal of Medicine, vol. 377, no. 1, pp. 28–40, Jul. 2017, doi: 10.1056/NEJMOA1700089.
L. Barbakadze, N. Kintraia, G. Burkadze, and S. Kepuladze, “ASSESSMENT OF PROLIFERATIVE ACTIVITY OF ENDOMETRIAL STROMAL TUMORS USING AgNOR TECHNOLOGY,” ქართველი მეცნიერები, vol. 5, no. 2, pp. 123–133, May 2023, doi: 10.52340/gs.2023.05.02.16.
T. Turashvili, G. Tevdorashvili, G. Burkadze, and S. Kepuladze, “Evaluation of proliferative activity of endometrial metaplasias by AgNor technology,” ქართველი მეცნიერები, vol. 5, no. 3, pp. 10–20, Jul. 2023, doi: 10.52340/2023.05.03.02.
B. Metreveli, D. Gagua, G. Burkadze, and S. Kepuladze, “Proliferative characteristics of eutopic and ectopic endometrium in adenomyosis using AgNOR technology,” Georgian Scientists, vol. 5, no. 1, pp. 59–71, Jan. 2023, doi: 10.52340/gs.2023.05.01.04.
S. Vannuccini, S. Clemenza, M. Rossi, and F. Petraglia, “Hormonal treatments for endometriosis: The endocrine background,” Rev Endocr Metab Disord, vol. 23, no. 3, pp. 333–355, Jun. 2022, doi: 10.1007/S11154-021-09666-W.
P. T. K. Saunders and A. W. Horne, “Endometriosis: Etiology, pathobiology, and therapeutic prospects,” Cell, vol. 184, no. 11, pp. 2807–2824, May 2021, doi: 10.1016/J.CELL.2021.04.041.
E. Chantalat et al., “Estrogen Receptors and Endometriosis,” Int J Mol Sci, vol. 21, no. 8, p. 2815, Apr. 2020, doi: 10.3390/IJMS21082815.
S. E. Bulun, S. Yildiz, M. Adli, and J. J. Wei, “Adenomyosis pathogenesis: insights from next-generation sequencing,” Hum Reprod Update, vol. 27, no. 6, pp. 1086–1097, Nov. 2021, doi: 10.1093/HUMUPD/DMAB017.
J. Zhai, S. Vannuccini, F. Petraglia, and L. C. Giudice, “Adenomyosis: Mechanisms and Pathogenesis,” Semin Reprod Med, vol. 38, no. 2–03, p. 129, May 2020, doi: 10.1055/S-0040-1716687.
S. C. Steinbuch, A. M. Lüß, S. Eltrop, M. Götte, and L. Kiesel, “Endometriosis-Associated Ovarian Cancer: From Molecular Pathologies to Clinical Relevance,” Int J Mol Sci, vol. 25, no. 8, p. 4306, Apr. 2024, doi: 10.3390/IJMS25084306.
D. Maeda and I. M. Shih, “Pathogenesis and the role of ARID1A mutation in endometriosis-related ovarian neoplasms,” Adv Anat Pathol, vol. 20, no. 1, p. 45, Jan. 2013, doi: 10.1097/PAP.0B013E31827BC24D.
T. Akahane, A. Sekizawa, Y. Purwosunu, M. Nagatsuka, and T. Okai, “The role of p53 mutation in the carcinomas arising from endometriosis,” Int J Gynecol Pathol, vol. 26, no. 3, pp. 345–351, Jul. 2007, doi: 10.1097/PGP.0B013E31802B41A8.
Copyright (c) 2025 Georgian Scientists

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.