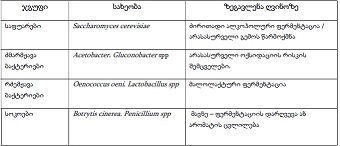
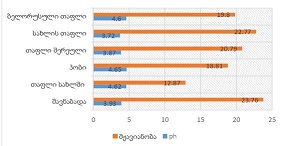
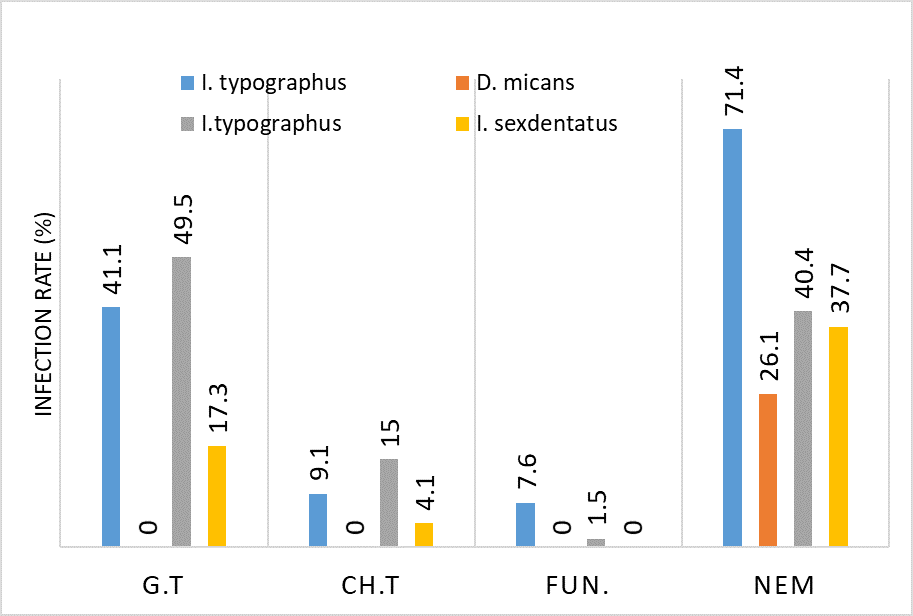
Phenylglycosylamines in Melanoidin Reaction
Downloads
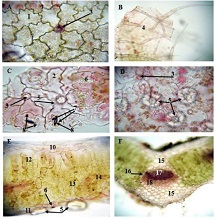
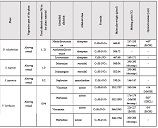
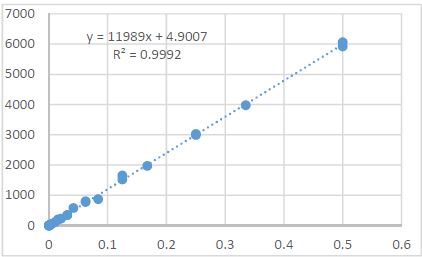
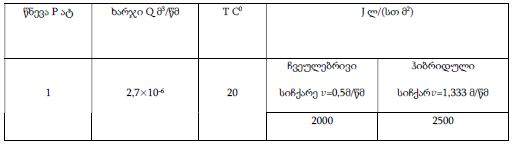
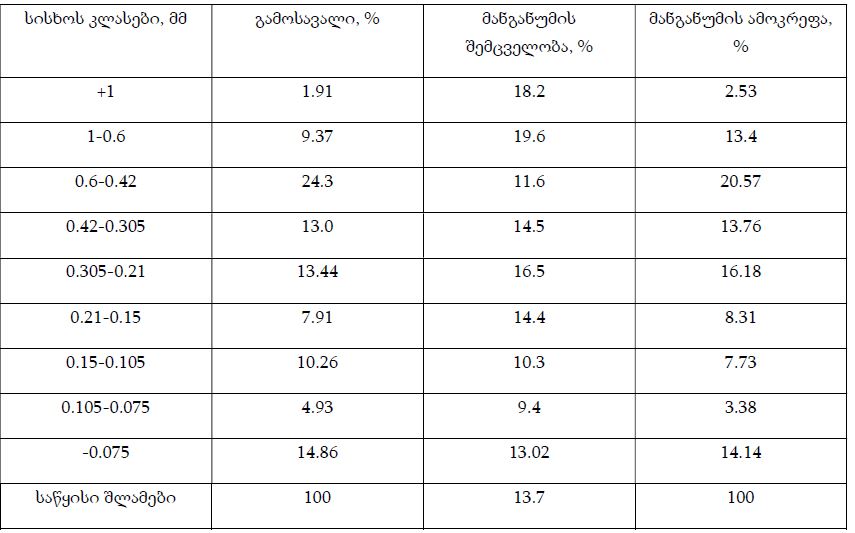
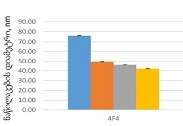
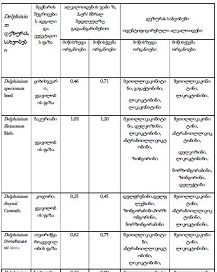
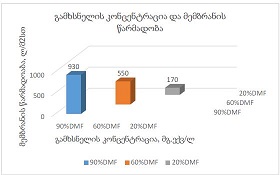
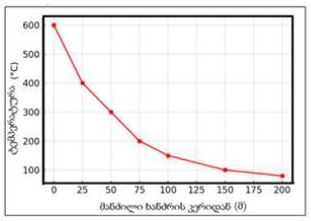
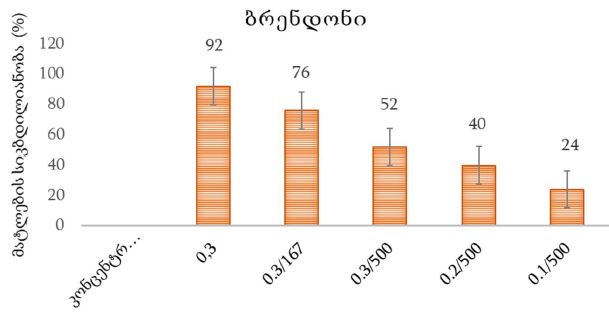
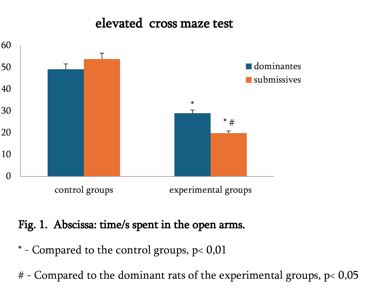
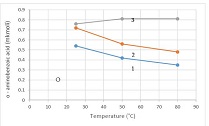
By the example of N-carboxy-phenylglycosylamines and N-hydroxy-phenylglycosylamines we have studied the dependence of transformation of N-glucoside – a key product of Maillard reaction on reaction medium pH and temperature [1,2]. We have determined the intensity of the mentioned aryl-N-glucosamines’ transformation into melanoidin according to number of formed melanoidin complexes (λ =470), and we have compared this process to melanoidin formation from respective amine component and glucose. We have conducted reaction in phosphate buffer medium, at following pH values: 5,0; 7,0; 9,2; at 1000°C during 2 hours, concentration of aryl-N-glucosamines or respective sugar-amine reacting mixture was 0.1 mole/l. Melanoidin reaction rate has been determined according to extinction of the formed melanoidin pigment (λ=470 nm). According to our experiment, N-glucosides and their respective sugar-amine mixtures participate in melanoidin reaction in a similar fashion: they generate melanoidin with roughly similar intensity at identical pH. At that, some difference has been noted between aminobenzoic acids and aminophenols; in particular, N-hydroxy-phenylglycosylamines and their respective sugar-amine mixtures intensively generate melanoidin pigments at high pH (9,2) and melanoidin reaction intensity reduces with increase of acidity. This fact makes aminophenols similar to aliphatic amino acids, which exactly in alkaline medium take more active part in melanoidin reaction [3].
Downloads
Sopromadze T. T., Kublashvili R. I. On the participation of aromatic amino acids in the process of melanoidin formation. Problems of Theoretical and Experimental Chemistry Abstracts of Reports, Ekaterinburg, 2002, p. 221.1.
Kublashvili R., Sophromadze T. Synthesis of N-glucosides of Amino Benzoooic Acids and Amino Phenols and Their Transformation into Melanoidin. – Bull. Georgian Acad. Sci., 2003, 167, 78-80.
Tserodze N., Tatiashvili L., Tsiskarishvili R., Dolidze L., Bolkvadze N., Karkashadze N., Uridia R.. Fraction composition of melanoidin pigment. Georgian Scientises. Vol.7 Issue4, 2005 https://doi.org/10.52340/gs.2025.07.04.13. p. 241-247.
Belitz H.D., Grosch W. food Chemistry. Berlin, Heidelberg, New York. Springer-Verlag. 1999, 263-318.
Ikan R. The Maillard Reaction. Chichester, John Wiley and Sons, 1996, 7-29.
Yaylayan V.A., Huyghues-desppointes A. Chemistry of Amadori Rearrangement Products: Analysis, Synthesis, Kinetics, Reactions, and Spectroscopic Properties. Crit. Rev. Food Sci. Nutr., 1994, 34, 321-369.
Kublashvili R., Chachua L., Samsonia Sh. Participation of free and N-glucosylated aminobenzoic acids in the Maillard reaction. Proc. Georg. Acad. Sci., 2005, 31, 64-69.
Kublashvili R. I., Ugrekhelidze D. Sh. On the participation of N-carboxyaenyl glucosylamines in the melanoidin reaction. Communications of the Academy of Sciences of the Georgian SSR, 1979, 93, 457-459.
Albert A., Sergent E. Ionization constants of acids and bases. M.L., Chemistry, 1964, 133-144 p.
Copyright (c) 2026 Georgian Scientists

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.