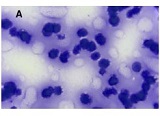
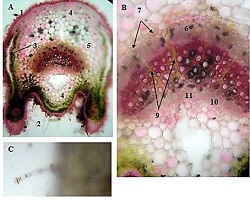
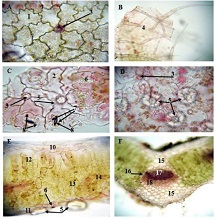
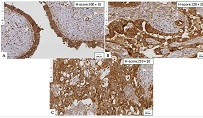
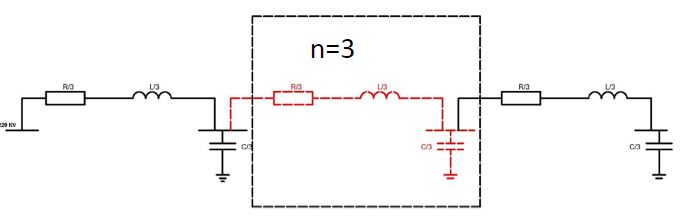
ASSESSMENT OF PROLIFERATIVE ACTIVITY OF ENDOMETRIAL STROMAL TUMORS USING AgNOR TECHNOLOGY
Downloads
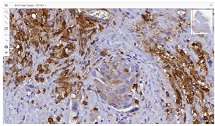
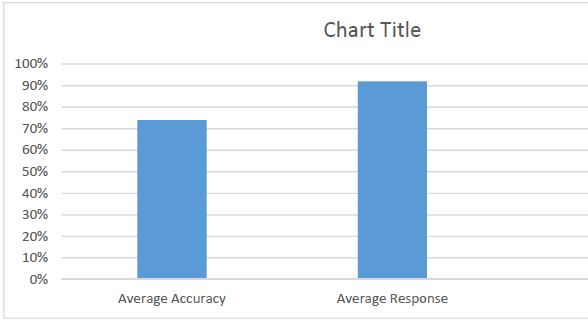
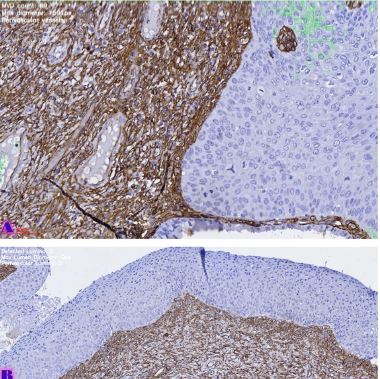
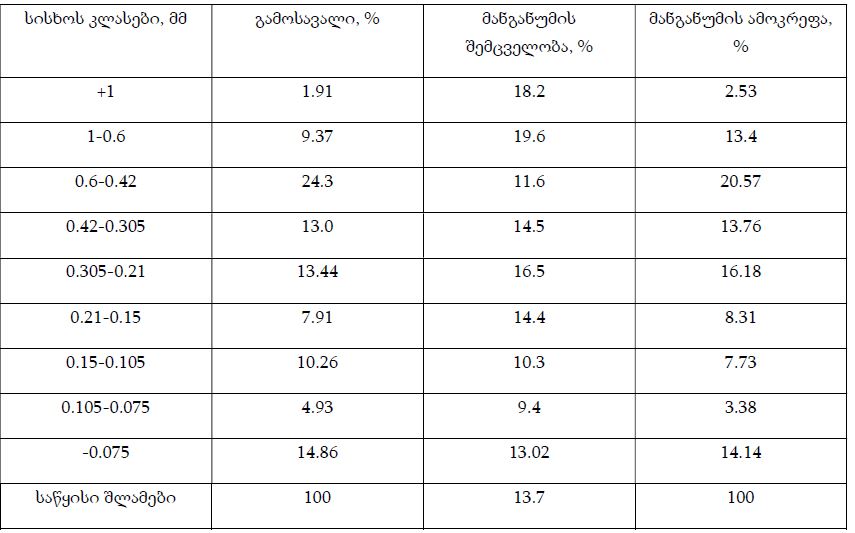
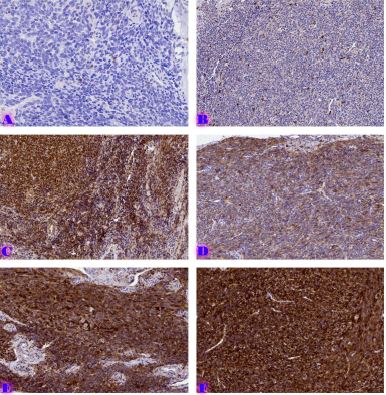
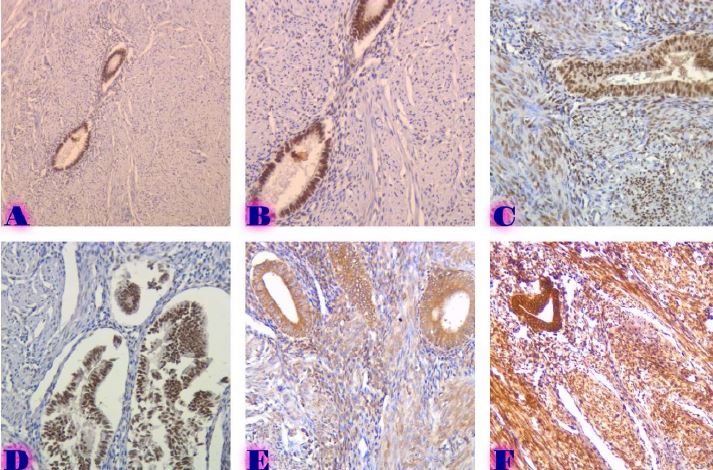
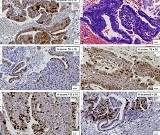
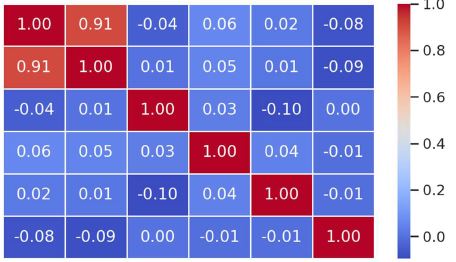
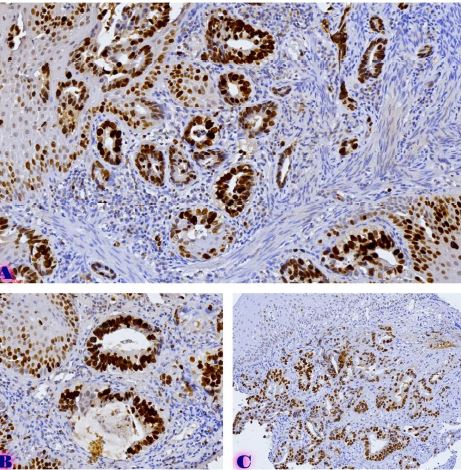
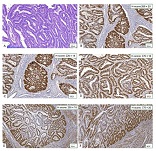
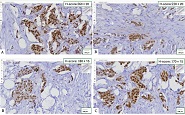
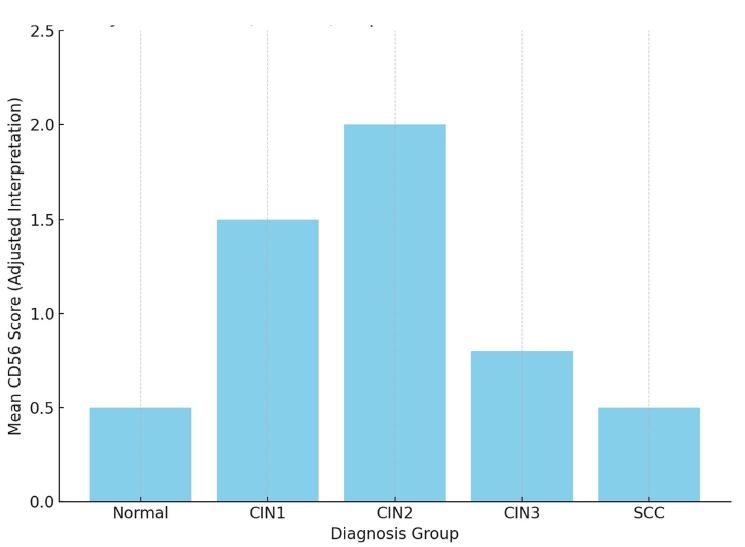
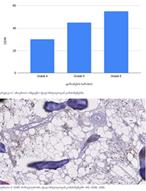
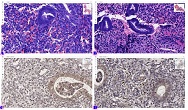
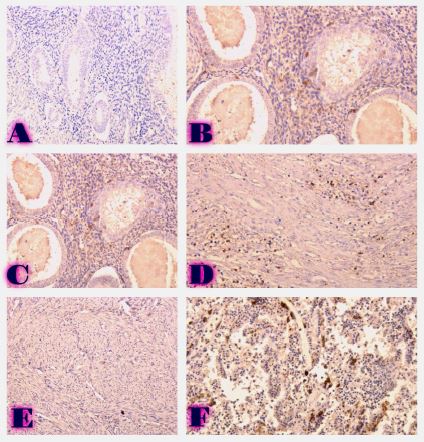
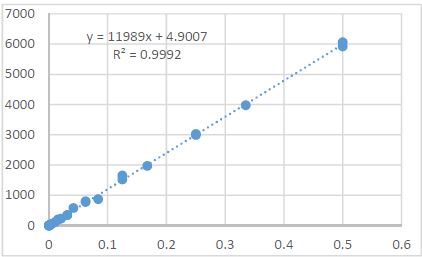
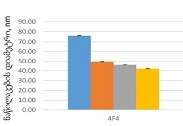
Endometrial stromal sarcomas (ESS) are rare neoplasms that arise from the endometrial stroma and account for less than 1% of all uterine malignancies. Diagnosis of ESS can be challenging due to its morphological overlap with other endometrial tumors, such as endometrial stromal nodules and stromal endometriosis. AgNOR count is a marker of cell proliferation and has been proposed as a potential tool for differentiating between different types of endometrial tumors.In this study, we analyzed the AgNOR count in 50 cases of endometrial tumors, including endometrial stromal nodules, stromal endometriosis, low-grade ESS, and high-grade ESS. Our results show that there is a significant difference in the AgNOR count across different types of endometrial tumors, with the mean AgNOR count increasing with the severity of the tumor. Endometrial stromal nodules had the lowest mean AgNOR count, while high-grade ESS had the highest.These findings suggest that AgNOR count may be a useful marker for differentiating between different types of endometrial tumors, including ESS. However, further research will be needed to confirm these findings and explore the clinical utility of this marker in the diagnosis and management of these conditions. Overall, our study contributes to our understanding of the biology of endometrial tumors and provides valuable information that may be useful in the development of diagnostic and treatment strategies for these conditions.
Downloads
B. Metreveli, D. Gagua, G. Burkadze, and S. Kepuladze, “PROLIFERATIVE CHARACTERISTICS OF EUTOPIC AND ECTOPIC ENDOMETRIUM IN ADENOMYOSIS USING AgNOR TECHNOLOGY,” GEORGIAN SCIENTISTS, vol. 5, no. 1, pp. 59–71, Jan. 2023, doi: 10.52340/gs.2023.05.01.04.
H. Sung et al., “Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries,” CA Cancer J Clin, vol. 71, no. 3, pp. 209–249, May 2021, doi: 10.3322/CAAC.21660.
F. Bray, J. Ferlay, I. Soerjomataram, R. L. Siegel, L. A. Torre, and A. Jemal, “Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries,” CA Cancer J Clin, vol. 68, no. 6, pp. 394–424, Nov. 2018, doi: 10.3322/CAAC.21492.
J. Ferreira, A. Félix, J. K. Lennerz, and E. Oliva, “Recent advances in the histological and molecular classification of endometrial stromal neoplasms,” Virchows Archiv, vol. 473, no. 6, pp. 665–678, Dec. 2018, doi: 10.1007/S00428-018-2470-6.
A. Talhouk et al., “Molecular subtype not immune response drives outcomes in endometrial carcinoma,” Clinical Cancer Research, vol. 25, no. 8, pp. 2537–2548, Apr. 2019, doi: 10.1158/1078-0432.CCR-18-3241.
S. Rousset-Rouviere et al., “Endometrial Carcinoma: Immune Microenvironment and Emerging Treatments in Immuno-Oncology,” Biomedicines, vol. 9, no. 6, Jun. 2021, doi: 10.3390/BIOMEDICINES9060632.
G. Puliyath and M. K. Nair, “Endometrial stromal sarcoma: A review of the literature,” Indian Journal of Medical and Paediatric Oncology, vol. 33, no. 01, pp. 1–6, Jan. 2012, doi: 10.4103/0971-5851.96960:
C. Lan, X. Huang, S. Lin, M. Cai, and J. Liu, “Endometrial Stromal Sarcoma Arising from Endometriosis: A Clinicopathological Study and Literature Review,” Gynecol Obstet Invest, vol. 74, no. 4, pp. 288–297, 2012, doi: 10.1159/000341706.
C. G. Peña et al., “LKB1 loss promotes endometrial cancer progression via CCL2-dependent macrophage recruitment,” J Clin Invest, vol. 125, no. 11, pp. 4063–4076, Nov. 2015, doi: 10.1172/JCI82152.
J. R. Goldblum, P. B. Clement, and W. R. Hart, “Adenomyosis with sparse glands: A potential mimic of low-grade endometrial stromal sarcoma,” Am J Clin Pathol, vol. 103, no. 2, pp. 218–223, 1995, doi: 10.1093/AJCP/103.2.218.
X. Matias-Guiu, B. Davidson, and S. F. Lax, “Can the classification of low-grade endometrial stromal tumors still be improved?,” Virchows Archiv, vol. 473, no. 6, pp. 663–664, Dec. 2018, doi: 10.1007/S00428-018-2474-2.
V. Krishnan, B. Schaar, S. Tallapragada, and O. Dorigo, “Tumor associated macrophages in gynecologic cancers,” Gynecol Oncol, vol. 149, no. 1, pp. 205–213, Apr. 2018, doi: 10.1016/J.YGYNO.2018.01.014.
X. Shi, J. Wang, Y. Lei, C. Cong, D. Tan, and X. Zhou, “Research progress on the PI3K/AKT signaling pathway in gynecological cancer (Review),” Mol Med Rep, vol. 19, no. 6, pp. 4529–4535, Jun. 2019, doi: 10.3892/MMR.2019.10121/HTML.
T. R. Soong et al., “Evidence for lineage continuity between early serous proliferations (ESPs) in the Fallopian tube and disseminated high‐grade serous carcinomas,” J Pathol, vol. 246, no. 3, pp. 344–351, Nov. 2018, doi: 10.1002/path.5145.
F. Rödel et al., “The Prognostic Relevance of the Proliferation Markers Ki-67 and Plk1 in Early-Stage Ovarian Cancer Patients With Serous, Low-Grade Carcinoma Based on mRNA and Protein Expression,” Front Oncol, vol. 10, Oct. 2020, doi: 10.3389/fonc.2020.558932.
N. Tavdgiridze, G. Tevdorashvili, S. Kepuladze, and G. Burkadze, “ASSESSMENT OF PROLIFERATIVE ACTIVITY OF IMMATURE OVARIAN TERATOMAS USING AgNOR TECHNOLOGY,” GEORGIAN SCIENTISTS, vol. 5, no. 1, pp. 233–248, Mar. 2023, doi: 10.52340/gs.2023.05.01.20.
P. Meshveliani, G. Didava, G. Tomadze, S. Kepuladze, and G. Burkadze, “EVALUATION OF PROLIFERATIVE ACTIVITY OF PRE-TUMOR AND TUMOR PROCESSES OF BARRETT’S ESOPHAGUS USING AGNOR TECHNOLOGY,” GEORGIAN SCIENTISTS, vol. 5, no. 2, pp. 49–62, Apr. 2023, doi: 10.52340/gs.2023.05.02.07.
Copyright (c) 2023 GEORGIAN SCIENTISTS

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.