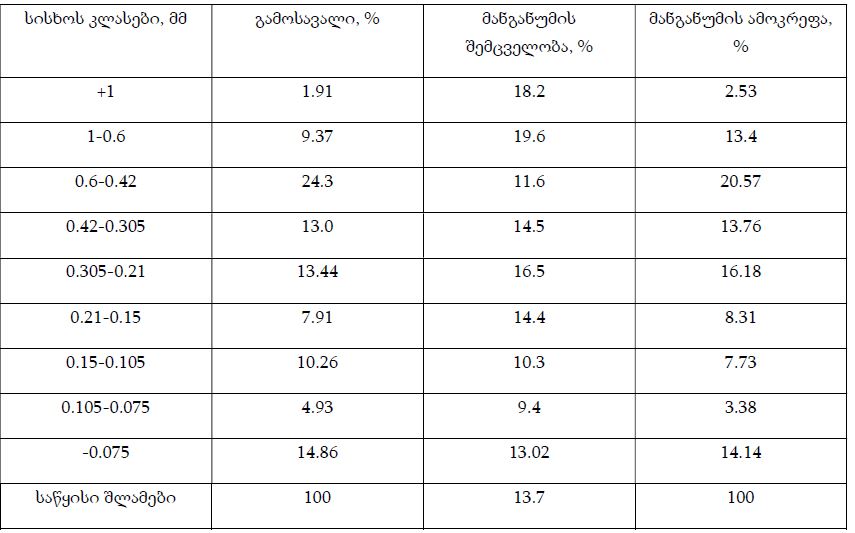
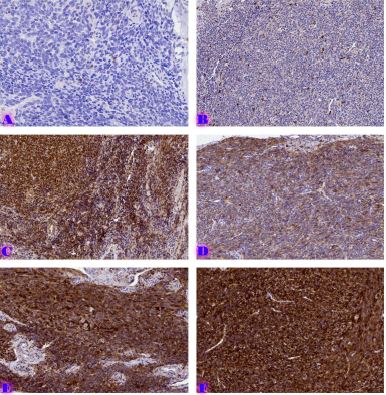
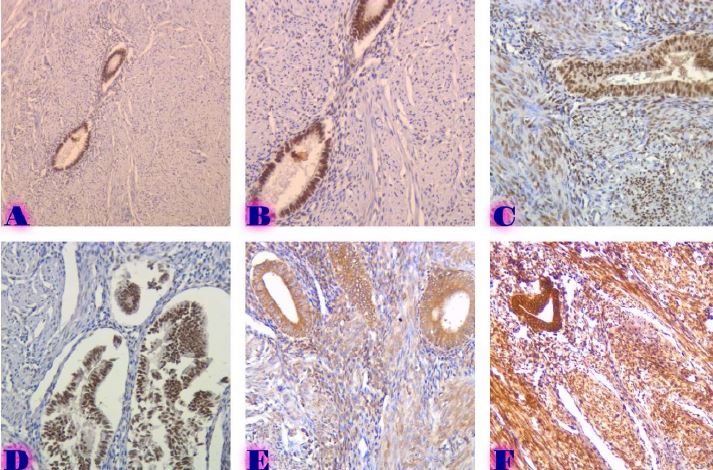
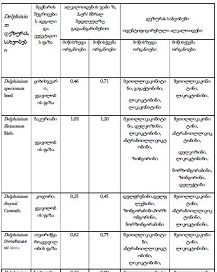
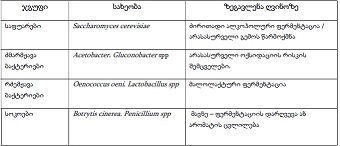
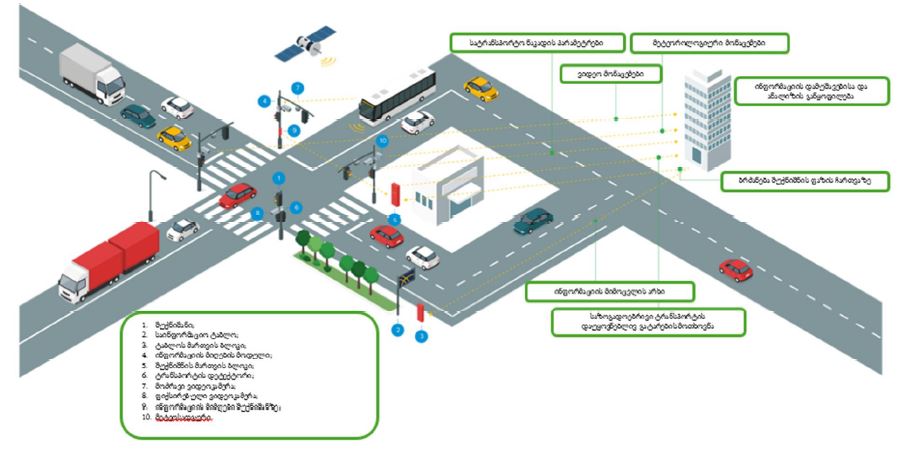
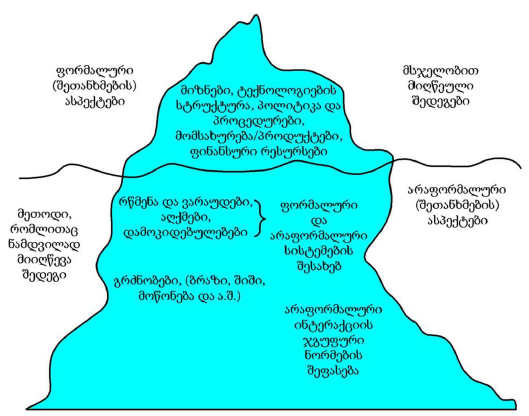
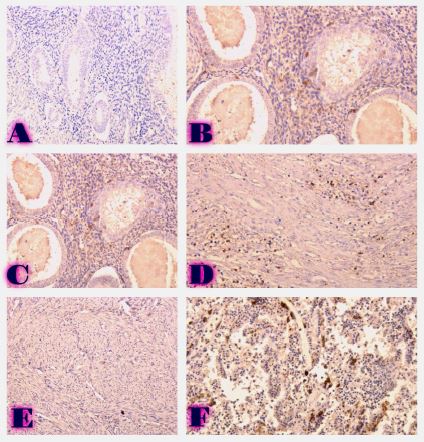
Features of Melatonin Receptor Expression in Endometrial Precancerous and Neoplastic Processes

Загрузки
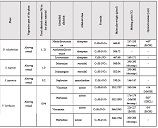
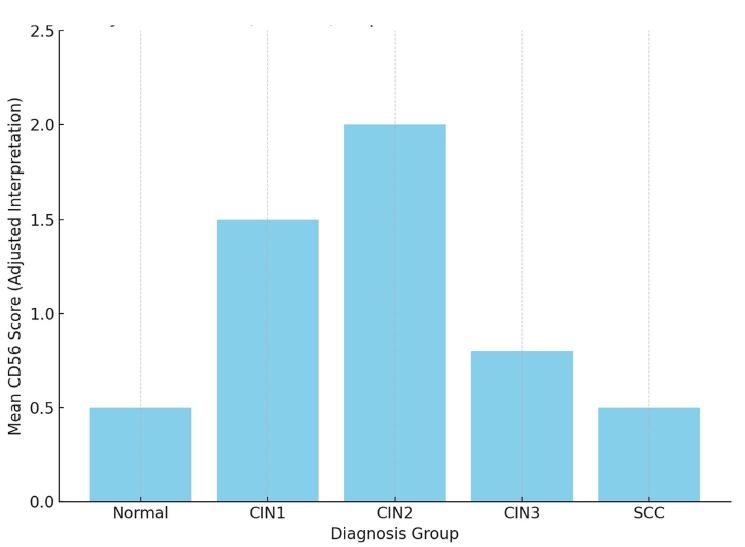
Melatonin is a multifunctional neuroendocrine hormone produced primarily by the pineal gland and widely recognized for its role in the regulation of circadian rhythms. In addition to its chronobiological function, melatonin exerts a variety of biological effects, including antioxidant activity, immune modulation, regulation of cell proliferation, and induction of apoptosis. These properties have attracted increasing attention in oncology research, particularly in hormone-dependent malignancies. Endometrial carcinoma is one of the most common gynecological cancers worldwide and often develops through a multistep process involving endometrial hyperplasia and progressive molecular changes in endometrial tissue. The pathogenesis of endometrial tumors is strongly influenced by hormonal regulation, particularly through estrogen and progesterone signaling pathways. However, emerging evidence suggests that additional hormonal regulators, including melatonin, may contribute to the complex mechanisms of endometrial carcinogenesis.
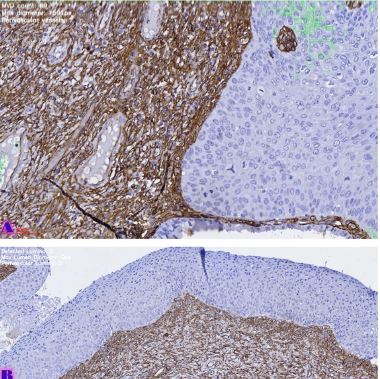
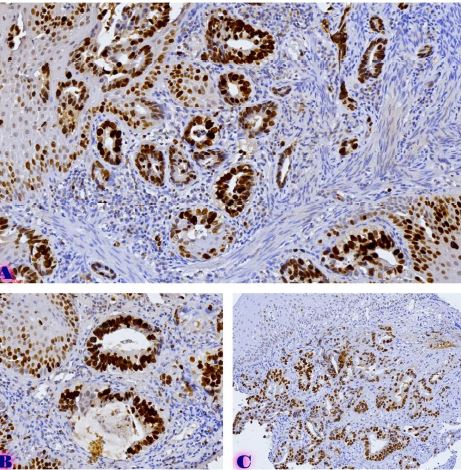
Melatonin exerts most of its biological effects through specific membrane receptors, mainly MT1 and MT2, which are expressed in many tissues, including the female reproductive tract. However, the expression patterns and biological significance of melatonin receptors in precancerous and malignant endometrial lesions are still poorly understood. Understanding these relationships may provide important insights into the molecular mechanisms of progression from endometrial hyperplasia to carcinoma. Furthermore, assessment of melatonin receptor expression at different stages of endometrial pathology may help identify novel biomarkers with diagnostic or prognostic value.
A review of the existing literature highlights the potential importance of melatonin receptor signaling in the development and progression of endometrial tumors.
Скачивания
Pandi-Perumal SR, Trakht I, Srinivasan V, Spence DW, Maestroni GJM, Zisapel N, et al. Physiological effects of melatonin: Role of melatonin receptors and signal transduction pathways. Prog Neurobiol. 2008 Jul;85(3):335–53. doi:10.1016/j.pneurobio.2008.04.001 PubMed PMID: 18571301.
Karasek M, Winczyk K. Melatonin in humans. Journal of Physiology and Pharmacology. 2006 Nov;57(SUPPL. 5):19–39. doi:10.1056/nejm199701163360306 PubMed PMID: 17218758.
Blask DE, Brainard GC, Dauchy RT, Hanifin JP, Davidson LK, Krause JA, et al. Melatonin-depleted blood from premenopausal women exposed to light at night stimulates growth of human breast cancer xenografts in nude rats. Cancer Res. 2005 Dec 1;65(23):11174–84. doi:10.1158/0008-5472.CAN-05-1945 PubMed PMID: 16322268.
Srinivasan V, Spence DW, Pandi-Perumal SR, Trakht I, Esquifino AI, Cardinali DP, et al. Melatonin, environmental light, and breast cancer. Breast Cancer Res Treat. 2008 Apr;108(3):339–50. doi:10.1007/s10549-007-9617-5 PubMed PMID: 17541739.
Cao Y, Zhang H, Chen X, Li C, Chen J. Melatonin: a natural guardian in cancer treatment. Front Pharmacol. 2025;16:1617508. doi:10.3389/fphar.2025.1617508 PubMed PMID: 40756978.
Yuan L, Collins AR, Dai J, Dubocovich ML, Hill SM. MT1 melatonin receptor overexpression enhances the growth suppressive effect of melatonin in human breast cancer cells. Mol Cell Endocrinol. 2002 Jun 28;192(1–2):147–56. doi:10.1016/S0303-7207(02)00029-1 PubMed PMID: 12088876.
Vijayalaxmi, Thomas CR, Reiter RJ, Herman TS. Melatonin: From basic research to cancer treatment clinics. Journal of Clinical Oncology. 2002 May 15;20(10):2575–601. doi:10.1200/JCO.2002.11.004 PubMed PMID: 12011138.
Del Río B, Garćia Pedrero JM, Martínez-Campa C, Zuazua P, Lazo PS, Ramos S. Melatonin, an endogenous-specific inhibitor of estrogen receptor α via calmodulin. Journal of Biological Chemistry. 2004 Sep 10;279(37):38294–302. doi:10.1074/jbc.M403140200 PubMed PMID: 15229223.
Reiter RJ, Sharma R, Rosales-Corral S. Anti-warburg effect of melatonin: A proposed mechanism to explain its inhibition of multiple diseases. Int J Mol Sci. 2021 Jan 2;22(2):1–24. doi:10.3390/ijms22020764 PubMed PMID: 33466614.
Barrenetxe J, Delagrange P, Martínez JA. Physiological and metabolic functions of melatonin. J Physiol Biochem. 2004;60(1):61–72. doi:10.1007/BF03168221 PubMed PMID: 15352385.
Travis RC, Allen DS, Fentiman IS, Key TJ. Melatonin and breast cancer: A prospective study. J Natl Cancer Inst. 2004 Mar 17;96(6):475–82. doi:10.1093/jnci/djh077 PubMed PMID: 15026473.
Dana PM, Sadoughi F, Mobini M, Shafabakhsh R, Chaichian S, Moazzami B, et al. Molecular and Biological Functions of Melatonin in Endometrial Cancer. Curr Drug Targets. 2019 Sep 27;21(5):519–26. doi:10.2174/1389450120666190927123746 PubMed PMID: 31560286.
Wurtman RJ, Axelrod J. The Formation, Metabolism, and Physiologic Effects of Melatonin. Adv Pharmacol. 1968;6(C):141–51. doi:10.1016/S1054-3589(08)61167-9 PubMed PMID: 4874443.
Kumari A, Quraishi R, Kumar A, Singh S, Singhal S, Singh N. Association of sleep quality and urinary melatonin levels with endometrial cancer: A case control pilot study. Ind Psychiatry J. 2026 Jan;35(1):173–5. doi:10.4103/ipj.ipj_222_25
Molecular and Biological Functions of Melatonin in Endometrial Cancer | Bentham Science Publishers [Internet]. [cited 2026 Mar 16]. Available from: https://www.benthamdirect.com/content/journals/cdt/10.2174/1389450120666190927123746
Melatonin alleviates progression of uterine endometrial cancer by suppressing estrogen/ubiquitin C/SDHB-mediated succinate accumulation - ScienceDirect [Internet]. [cited 2026 Mar 16]. Available from: https://www.sciencedirect.com/science/article/abs/pii/S0304383520300719
Grin W, Grünberger W. A significant correlation between melatonin deficiency and endometrial cancer. Gynecol Obstet Invest. 1998;45(1):62–5. doi:10.1159/000009926 PubMed PMID: 9473168.
Gu C, Yang H, Chang K, Zhang B, Xie F, Ye J, et al. Melatonin alleviates progression of uterine endometrial cancer by suppressing estrogen/ubiquitin C/SDHB-mediated succinate accumulation. Cancer Lett. 2020 Apr 28;476:34–47. doi:10.1016/j.canlet.2020.02.009 PubMed PMID: 32061949.
Nagata C, Nagao Y, Yamamoto S, Shibuya C, Kashiki Y, Shimizu H. Light exposure at night, urinary 6-sulfatoxymelatonin, and serum estrogens and androgens in postmenopausal Japanese women. Cancer Epidemiology Biomarkers and Prevention. 2008 Jun;17(6):1418–23. doi:10.1158/1055-9965.EPI-07-0656 PubMed PMID: 18559557.
Cos S, González A, Güezmes A, Mediavilla MD, Martínez-Campa C, Alonso-González C, et al. Melatonin inhibits the growth of DMBA-induced mammary tumors by decreasing the local biosynthesis of estrogens through the modulation of aromatase activity. Int J Cancer. 2006 Jan 15;118(2):274–8. doi:10.1002/ijc.21401 PubMed PMID: 16080194.
Kepuladze S, Burkadze G, Kokhreidze I, Kepuladze S, Burkadze G, Kokhreidze I. Epithelial-Mesenchymal Transition Indexes in Triple-Negative Breast Cancer Progression and Metastases. Cureus. 2024 Sep 6;16(9). doi:10.7759/CUREUS.68761
Shengelaia K, Kokhreidze I, Burkadze G, Kepuladze S, Tevzadze N. Challenges of Androgen Receptor Expression in Epithelial Tumors of the Breast and Ovary. ქართველი მეცნიერები. 2023 Aug 4;5(3):75–84. doi:10.52340/2023.05.03.09
Turashvili T, Tevdorashvili G, Burkadze G, Kepuladze S. Evaluation of proliferative activity of endometrial metaplasias by AgNor technology. ქართველი მეცნიერები. 2023 Jul 9;5(3):10–20. doi:10.52340/2023.05.03.02
Dunn GP, Koebel CM, Schreiber RD. Interferons, immunity and cancer immunoediting. Nat Rev Immunol. 2006 Nov 13;6(11):836–48. doi:10.1038/nri1961 PubMed PMID: 17063185.
Rousset-Rouviere S, Rochigneux P, Chrétien AS, Fattori S, Gorvel L, Provansal M, et al. Endometrial Carcinoma: Immune Microenvironment and Emerging Treatments in Immuno-Oncology. Biomedicines. 2021 Jun 1;9(6). doi:10.3390/BIOMEDICINES9060632 PubMed PMID: 34199461.
Rousset-Rouviere S, Rochigneux P, Chrétien AS, Fattori S, Gorvel L, Provansal M, et al. Endometrial Carcinoma: Immune Microenvironment and Emerging Treatments in Immuno-Oncology. Biomedicines. 2021 Jun 1;9(6):632. doi:10.3390/BIOMEDICINES9060632 PubMed PMID: 34199461.
Rousset-Rouviere S, Rochigneux P, Chrétien AS, Fattori S, Gorvel L, Provansal M, et al. Endometrial Carcinoma: Immune Microenvironment and Emerging Treatments in Immuno-Oncology. Biomedicines. 2021 Jun 1;9(6):632. doi:10.3390/BIOMEDICINES9060632 PubMed PMID: 34199461.
A Gonzalez-Gonzalez AGCAGJMMCMCSC. Complementary actions of melatonin on angiogenic factors, the angiopoietin/Tie2 axis and VEGF, in cocultures of human endothelial and breast cancer cells. Oncol Rep. 2018;39:433–41.
RJ Reiter SRCDTMJAGBX. Melatonin as a mitochondria-targeted antioxidant: one of evolution’s best ideas. Cell Mol Life Sci. 2017;74:3863–81.
Korkmaz A, Reiter RJ, Topal T, Manchester LC, Oter S, Tan DX. Melatonin: An established antioxidant worthy of use in clinical trials. Molecular Medicine. 2009 Jan;15(1–2):43–50. doi:10.2119/MOLMED.2008.00117 PubMed PMID: 19011689.
M Ortiz-Franco EPBQDACIRGE. Effect of melatonin supplementation on antioxidant status and DNA damage in high intensity trained athletes. Int J Sports Med. 2017;38:1117–25.
RS Barberino VMARRPXJJS. Melatonin protects against cisplatin-induced ovarian damage in mice via the MT1 receptor and antioxidant activity. Biol Reprod. 2017;96:1244–55.
Wellner U, Schubert J, Burk UC, Schmalhofer O, Zhu F, Sonntag A, et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat Cell Biol. 2009 Dec;11(12):1487–95. doi:10.1038/NCB1998 PubMed PMID: 19935649.
Kepuladze S, Nikolaishvili N, Chichua G, Burkadze G. Molecular markers of the progression of conjunctival neoplastic epithelial lesions and its correlation with P16 and HPV expression. Indian Journal of Pathology and Oncology. 2023 Mar 28;10(1):40–3. doi:10.18231/J.IJPO.2023.007
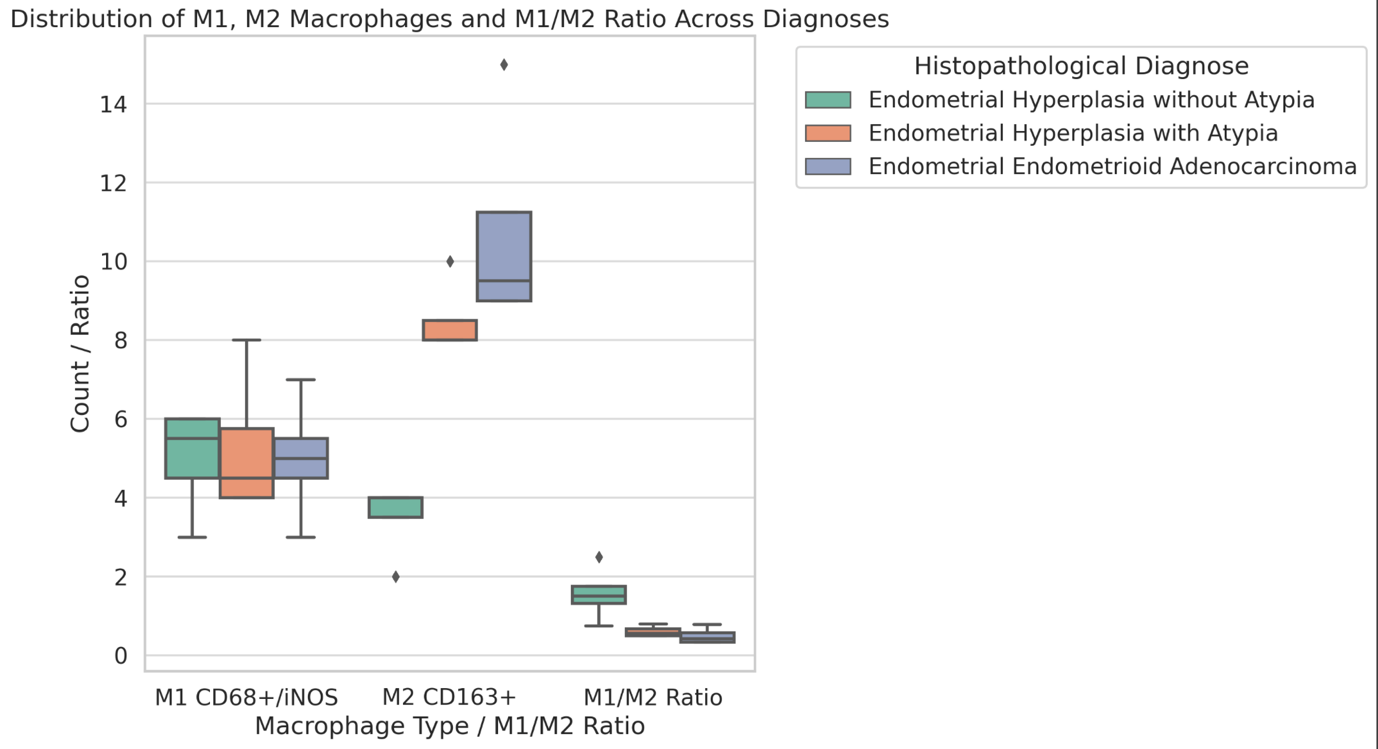
Kepuladze S, Devadze R, Gvenetadze A, Burkadze G. Distribution of tumor-associated macrophages and M1/M2 polarization in different types and grades of ovarian tumors. Indian Journal of Pathology and Oncology. 2022 Dec 28;9(4):318–21. doi:10.18231/J.IJPO.2022.076
Kepuladze S, Dzotsenidze T, Gvenetadze A, Gachechiladze M, Burkadze G. Immunohistochemical phenotype of fallopian tubes in patients with different grades of serous ovarian carcinoma. Indian Journal of Pathology and Oncology. 2022 Dec 28;9(4):301–5. doi:10.18231/J.IJPO.2022.073
Copyright (c) 2026 Georgian Scientists

Это произведение доступно по лицензии Creative Commons «Attribution-NonCommercial-NoDerivatives» («Атрибуция — Некоммерческое использование — Без производных произведений») 4.0 Всемирная.