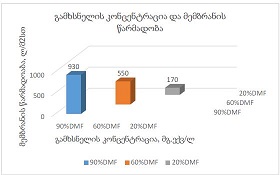
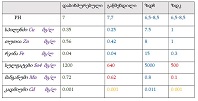
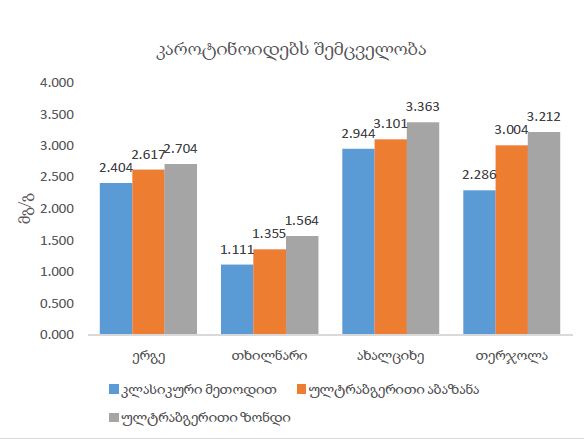
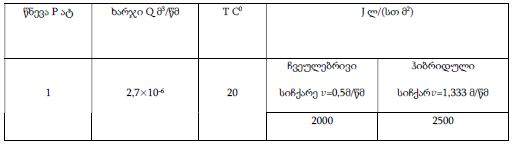
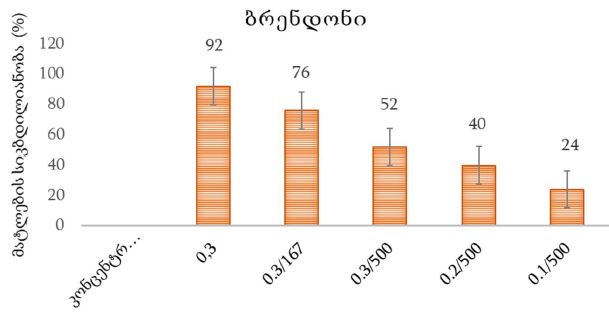
Concept for creating sunscreen cosmetics
Downloads
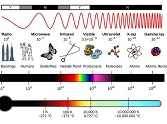
This paper discusses issues related to the problem of solar radiation. The possible consequences of irradiation with ultraviolet, visible and infrared areas of skin irradiation are considered. Based on literature data, the possibility of exposure to ultraviolet radiation (UV) is shown as one of the main risk factors for a number of human skin diseases. It should be noted that UV radiation can penetrate deeply into the epidermis and dermis, causing degradation of collagen and elastin fibers, and this is the main cause of oncological diseases such as melanoma, associated with DNA mutation, suppression of the antitumor immune response and increased skin inflammation. Sunburn is associated with a risk of melanoma, basal cell and squamous cell carcinomas.Visible light is the result of a number of skin diseases Solar urticaria, Porphyrias like acute and cutaneous porphyrias. It should be noted that one type of porphyria is a consequence of chronic inflammation of the liver and iron overload of the liver; chronic actinic dermatitis and systemic lupus erythematosus, melasma and skin aging are also associated with the visible light. Disturbances in the nature of the skin condition are also recorded when they are exposed to the infrared (760–5000 nm) part of the solar spectrum. Even with weak irradiation with single or fairly small repeated doses of infrared radiation, free radicals appear. This paper describes the influence of components of the solar spectrum on the human body. Based on the literature review, the appearance and composition of the skin is affected by all three types of solar radiation - ultraviolet radiation with wavelengths from 100 to 400 nm, the visible region 400-760 nm and the near-infrared region 760 nm and above. All these types of radiation can cause not only sunburn, but also the possibility of cancer, skin aging and a number of other skin diseases. There are no universal means of protection. Apparently, given this fact and possible damage to the body, it is necessary to intensify scientific research on the development of external means of protection (ointments, creams, lotions, etc.) using substances of plant and natural origin with a low degree of toxicity.
Downloads
Li Anqi , Li Shiqin, Tang Jie, Xiong of Lidan.Natural Ingredients in Sunscreens: Topical Sunscreen (SPF) Formulations. Biomedicine and Pharmacotherapy. 2021 February; Volume 134, 111161.
Shang-Ru Tsai, Rui Yin, Ying-Ying Huang, Bor-Ching Sheu, Si-Chen Lee, and Michael R. Hamblin. Low-Level Light Therapy Potentiates NPe6-mediated Photodynamic Therapy in a Human Osteosarcoma Cell Line via Increased ATP. Photodiagnosis Photodyn Ther. 2015 Mar; 12(1): 123–130.
Daniel Barolet, François Christiaens, Michael R. Hamblin. Infrared and Skin: Friend or Foe. J Photochem Photobiol B. 2016 Feb; 155: 78–85.
Soyun Cho, Mi Hee Shin, Yeon Kyung Kim, Jo-Eun Seo, Young Mee Lee .Effects of infrared radiation and heat on human skin aging in vivo. J. Investig Dermatol Symp. Proc. 2009 Aug;14(1):15-9.
Maxim E. Darvin, Wolfram Sterry, Juergen Lademann, Theognosia Vergou . The Role of Carotenoids in Human Skin Molecules 2011, 16(12), 10491-10506.
Darwin M.E. et al. Intravital Raman spectroscopic analysis of the effect of IR radiation on the carotenoid antioxidant substances beta-carotene and lycopene in human skin. Formation of free radicals. Letters on laser physics. 2007; 4(4):318–321.
R M Lucas , S Yazar , A R Young , M Norval ,, F R de Gruijl , and andere . Human health in relation to exposure to solar ultraviolet radiation in a changing stratospheric ozone and climate. Photochemical and Photobiological Sciences, 2019, 18(3):641-680.
Ghiasvand, R., Robsahm, T. E., Green, A. C., Rueegg, C. S., Weiderpass, and andere. Association of phenotypic characteristics and UV radiation exposure with risk of melanoma on different body sites. JAMA Dermatology, 2018,155(1), 39–49.
Arnold M., de Vries E., Whiteman D. C., Jemal A., Bray F., and andere. Global burden of cutaneous melanoma attributable to ultraviolet radiation in 2012. International Journal of Cancer, 2019,143(6), 1305–1314.
O’Sullivan, D. E., Brenner, D. R., Villeneuve, P. J., Walter, S. D., Demers, P. A., and andere. Estimates of the current and future burden of melanoma attributable to ultraviolet radiation in Canada. Preventive Medicine, 2019, 122,81- 90.
Little M. P., Linet M. S., Kimlin M. G., Lee T., Tatalovich Z., and andere. Cumulative solar ultraviolet radiation exposure and basal cell carcinoma of the skin in a nationwide US cohort using satellite and ground-based measures. Environmental Health, 2019,18(1), 114.
Kolitz E., Lopes Arffa M., Pineider J., Bogucka R., and andere.. UV exposure and the risk of keratinocyte carcinoma in skin of color: a systematic review. JAMA Dermatology, 2022, 158(5),542-546.
Olsen C. M., Pandeya, N., & Whiteman, D. C. International increases in Merkel cell carcinoma incidence rates between 1997 and 2016. Journal of Investigative Dermatology, 2021, 141(11), 2596-2601.
de Melo A. C., Santos Thuler L. C..Trends in the incidence and morbidity of Merkel cell carcinoma in Brazil. Future Oncology, 2021, 17(22), 2857–2865.
Stang A., Becker, J. C., Nghiem P., Ferlay J. The association between geographic location and incidence of Merkel cell carcinoma in comparison to melanoma: an international assessment. European Journal of Cancer, 2018, 94, 47–60.
Ferrándiz-Pulido C., Lera-Imbuluzqueta M., Ferrándiz C., Plazas-Fernandez M. J. (2018). Prevalence of actinic keratosis in different regions of Spain: the EPIQA Study. Actas Dermo-Sifiliográficas, 2018,109(1), 83–86.
Tokez S., Wakkee M., Louwman M., Noels E., Nijsten T., Hollestein L. (2020). Assessment of cutaneous squamous cell carcinoma (cSCC) in situ incidence and the risk of developing invasive cSCC in patients with prior cSCC in situ vs the general population in the Netherlands, 1989–2017. JAMA Dermatology, 2020, 156 (9), 973 –981.
Savoye I., Olsen C. M., Whiteman D. C., Bijon, A. and andere. Patterns of ultraviolet radiation exposure and skin cancer risk: the E3N-SunExp Study. Journal of Epidemiology, 2018, 28 (1), 27–33.
Kawai K., VoPham T., Drucker A., Curhan S. G., Curhan G. C. Ultraviolet radiation exposure and the risk of Herpes zoster in three prospective cohort studies. Mayo Clinic Proceedings, 2020, 95 (2), 283 – 292.
Tripathi R., Mazmudar R. S., Knusel K. D.,and andere. Trends in emergency department visits due to sunburn and factors associated with severe sunburns in the United States. Archives of Dermatological Research, 2021, 313(2), 79 -88.
Sarkar M. K., Hile G. A., Tsoi L. C., Xing, X., and andere. Photosensitivity and type I IFN responses in cutaneous lupus are driven by epidermal-derived interferon kappa. Annals of Rheumatic Diseases, 2018, 77(11), 1653–1664.
Gutierrez D., Gaulding J. V., Motta Beltran A. F., Lim H. W., Pritchett E. N. Photodermatoses in skin of colour. Journal of the European Academy of Dermatology and Venereology, 2018,32 (11), 1879–1886.
Gether L., Overgaard L. K., Egeberg A., Thyssen J. P. Incidence and prevalence of rosacea: a systematic review and meta-analysis. British Journal of Dermatology, 2018, (2), 282 - 289.
Rutter K. J., Ashraf I., Cordingley L., Rhodes L. E. Quality of life and psychological impact in the photodermatoses: a systematic review. British Journal of Dermatology, 2020, 182(5), 1092–1102.
Lucas R. M., Rodney Harris R. On the nature of evidence and ‘proving’ causality: smoking and lung cancer vs. sun exposure, vitamin D and multiple sclerosis. International Journal of Environmental Research and Public Health, 2018, 15 (8),172- 1726.
Yaar M, Gilchrest BA. Photoageing: mechanism, prevention and therapy. Br J Dermatol. 2007;157:874–887.
Sachs DL, Varani J, Chubb H, Fligiel SEG, Cui Y, Calderone K, et al. Atrophic and hypertrophic photoaging: clinical, histologic, and molecular features of 2 distinct phenotypes of photoaged skin. J Am Acad Dermatol. 2019;81:480–488.
Poon F, Kang S, Chien AL. Mechanisms and treatments of photoaging. Photodermatol Photoimmunol Photomed. 2015;31:65–74.
Young AR, Claveau J, Rossi AB. Ultraviolet radiation and the skin: photobiology and sunscreen photoprotection. J Am Acad Dermatol. 2017;76:S100–S109.
Zhang J.A., Yin Z., Ma L.V. et al. Protective effect of baicalin against UV-B induced photoaging: an in vitro and in vivo study. Flat one. 2014; 9(6):e99703.
Ryu J., Pak S.J., Kim I.H., Choi Y.H., Nam T.J. Protective effect of porphyry-334 on UVA-induced photoaging in human skin fibroblasts. Int J Mol Med. 2014; 34(3): 796-803.
Krystyna Joanna,Gromkowska Kępka, Anna Puścion Jakubik, Renata Markiewicz Żukowska, Katarzyna Socha. The impact of ultraviolet radiation on skin photoaging - review of in vitro studies. J Cosmet Dermatol. 2021 Nov;20(11):3427-3431.
Diffey B. Solar spectral irradiance and summary outputs using Excel. Photochem Photobiol. 2015;91:553 - 557.
Bennet D, Viswanath B, Kim S, An JH. An ultra sensitive biophysical risk assessment of light effect on skin cells. Oncotarget. 2017;8(29):47861‐ 47875.
Masson‐Meyers DS, Bumah VV, Enwemeka CS. A comparison of four methods for determining viability in human dermal fibroblasts irradiated with blue light. J Pharmacol Toxicol Methods. 2016;79:15‐22.
Lohan S, Müller R, Albrecht S, et al. Free radicals induced by sunlight in different spectral regions ‐ in vivo versus ex vivo study. Exp Dermatol. 2016;25:380‐385.
Balic A, Mokos M. Do we utilize our knowledge of the skin protective effects of carotenoids enough? Antioxidants. 2019, 8, 259.
Choe CS, Ri JR, Schleusener J, Lademann J, Darvin ME. The non‐homogenous distribution and aggregation of carotenoids in the stratum corneum correlates with the organization of intercellular lipids in vivo. Exp Dermatol. 2019:1‐7. 10.1111/exd.14018.
Mendrok‐Edinger C, Campice R, Gadsinski K, Schuetz R. Into the blue ‐ novel test reveals blue light damage, protection strategies. Cosmet Toiletr. 2018;133(1):13‐29.
Vandersee S, Beyer M, Lademann J, Darvin ME. Blue‐violet light irradiation dose dependently decreases carotenoids in human skin, which indicates the generation of free radicals. Oxid Med Cell Longev. 2015; 579675.
Haylett AK, Koumaki D, Rhodes LE. Solar urticaria in 145 patients: assessment of action spectra and impact on quality of life in adults and children. Photodermatol Photoimmunol Photomed. 2018;34:262-268.
. Du‐Tanh A, Debu A, Lalheve P, Guillot B, Dereure O, Peyron JL. Solar urticaria: a time‐extended retrospective series of 61 patients and review of literature. Eur J Dermatol. 2013;12:202-207.
Goetze S, Elsner P. Solar urticaria. J Dtsch Dermatol Ges. 2015;13(12):1250–1253.
Kishimoto I, Uetsu N, Tanimura H, Fujii H, Okamoto H. Solar urticaria with a wide action spectrum from UVB to visible light complicated with UVA induced polymorphous light eruption. Photodermatology, photoimmunology & photomedicine. 2017;33(3):172–175.
Hexsel CL, Hamzavi IH, Lim HW. Effects of visible light on the skin. Photochemistry and photobiology. 2008;84(2):450–462.
Balwani M, Desnick RJ. The porphyrias: advances in diagnosis and treatment. Blood 2012; 120: 4496–4504.
Besur S, Hou W, Schmeltzer P, Bonkovsky HL. Clinically important features of porphyrin and heme metabolism and the porphyrias. Metabolites 2014; 4: 977–1006.
Ramanujam VM, Anderson KE. Porphyria diagnostics‐part 1: a brief overview of the porphyrias. Curr Protoc Hum Genet 2015; 86: 17.20.11–17.20.26.
Artz CE, Farmer CM, Lim HW. Polymorphous Light Eruption: a Review. Current dermatology reports. 2019;8(3):110–116.
Pérez-Ferriols A, Barnadas M, Gardeazábal J, et al. Solar urticaria: epidemiology and clinical phenotypes in a Spanish series of 224 patients. Actas Dermo-Sifiliográficas (English Edition). 2017;108(2):132–139.
Silpa-archa N, Wongpraparut C, Leenutaphong V. Analysis of solar urticaria in Thai patients. Asian Pacific journal of allergy and immunology. 2016;34(2):146–152.
Quatrano NA, Shvartsbeyn M, Meehan SA, Soter NA, Cohen DE. Chronic actinic dermatitis occurring in an adult with atopic dermatitis. Dermatology online journal. 2015;21(12).
Van Weelden H, Velthuis PJ, Baart de la Faille H. Light-induced skin lesions in lupus erythematosus: photobiological studies. Archives of dermatological research. 1999;281(7):470–474.
Jang MS. Clinical and molecular analysis of the effects of visible light on melasma. Pacific Pharma Scholarsh Report-4. 2015;128-129.
Randhawa M, Seo I, Liebel F, Southall MD, Kollias N, Ruvolo E. Visible light induces melanogenesis in human skin through a photoadaptive response. PLoS One. 2015, 20:e0130949.
Dupont E, Gomez J, Bilodeau D. Beyond UV radiation: a skin under challenge. Int J Cosmet Sci. 2013;35:224-232.
Randhawa M, Seo I, Liebel F, Southall MD, Kollias N, Ruvolo E. Visible light induces melanogenesis in human skin through a photoadaptive response. PLoS One. 2015;20.
Nakashima Y, Ohta S, Wolf AM. Blue light induced oxidative stress in live skin. Free Rad Biol Med. 2017;108:300-310.
Martini APM, Maja Campos PMBG. Influence of visible light on cutaneous hyperchromias: clinical efficacy of broad spectrum sunscreens. Photodermatol Photoimmunol Photomed. 2018;34:241-248.
Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long wavelength ultraviolet al and visible light on pigmentation and erythema. Brit J Dermatol. 2018;178:1173-1180.
Kohli I, Nahhas AF, Braunberger TL, et al. Spectral characteristics of visible light induced pigmentation and visible light protection factor. Photodermatol Photoimmunol Photomed. 2019;1-7. 10.1111/phpp.12490.
Nahhas AF, Abdel Malek ZA, Kohli I, Braunberger TL, Lim HW, Hamzavi IH. The potential role of antioxidants in mitigating skin hyperpigmentation resulting from ultraviolet and visible light‐induced oxidative stress. Photodermatol Photoimmunol Photomed. 2018:1-9.
Robert C, Bonnet M, Marques S, Numa M, Doucet O. Low and moderate doses of IR A disrupt skin extracellular matrix homeostasis and promote skin photodamage. Skin Pharmacol Physiol. 2015,28, р.196–204.
Darwin M.E., Haag C., Meinke M., Zastrow L., Sterry W., Lademann J. Infrared A Radical Production in Human Tissues. Skin Pharmacol Physiol. 2010;23:40–6.
Nia, K., Maibach, H. Infrared Radiation: Mechanisms, Implications, and Protection. In: Farage, M., Miller, K., Maibach, H. (eds) Textbook of Aging Skin. Springer, Berlin, Heidelberg. (2015).
Еrnández E.·, Fajarí L. , Rodríguez G., Cócera M. , Moner V. И др. Reducing the Harmful Effects of Infrared Radiation on the Skin Using Bicosomes Incorporating β-Carotene. Skin Pharmacol Physiol, 2016;29, р.169-177.
Biniek K, Levi K, Dauskardt RH: Solar UV radiation reduces the barrier function of human skin. Proc Natl Acad Sci , 2012;109 ,Р.17111-17116.
Krutmann J, Morita A, Chung JH: Sun exposure: what molecular photodermatology tells us about its good and bad sides. J Invest Dermatol 2012;132:976-984.
Akhalaya MY, Maksimov G, Rubin A, Lademann J, Darvin M: Molecular action mechanisms of solar infrared radiation and heat on human skin. Ageing Res Rev 2014;16:1-11.
Copyright (c) 2023 GEORGIAN SCIENTISTS

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.