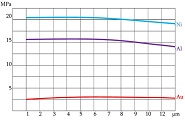
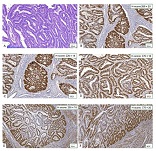
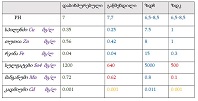
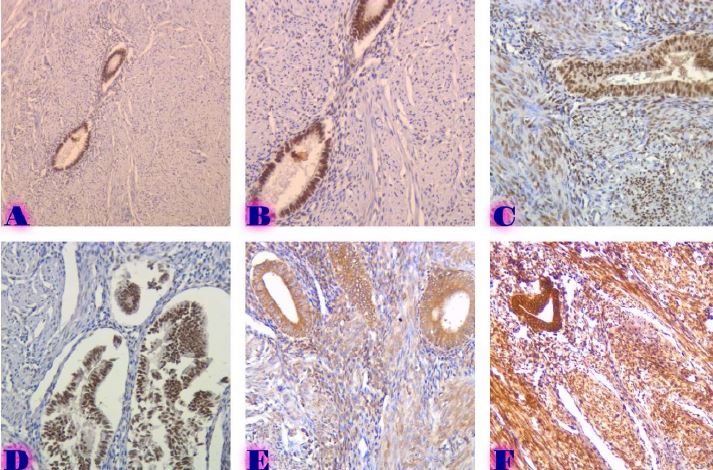
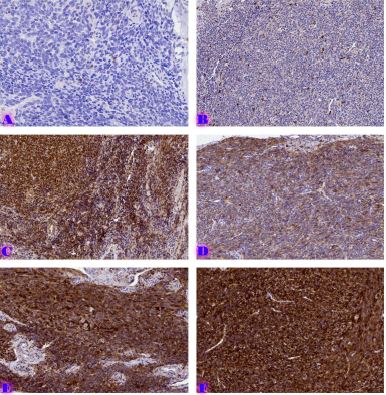
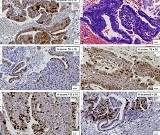
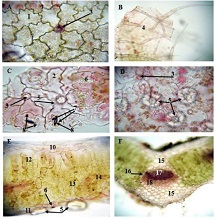
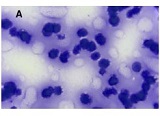
Proliferative-apoptotic, hormonal receptor, and immune response characteristics of rheumatoid arthritis
Downloads
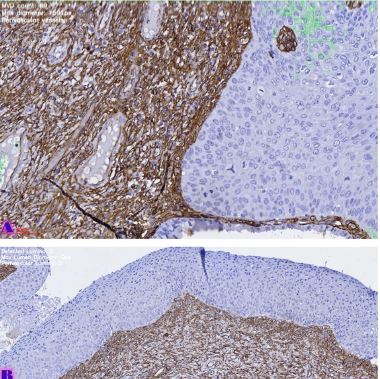
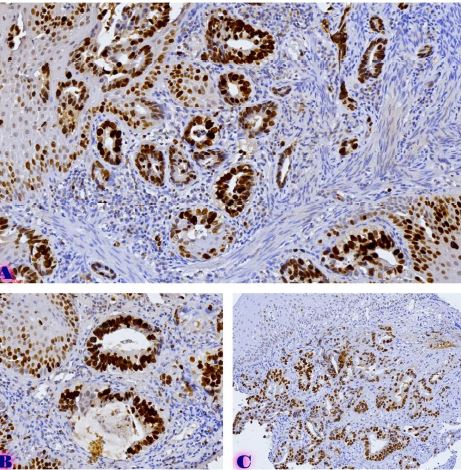
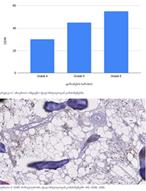
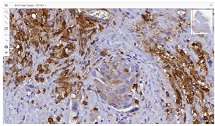
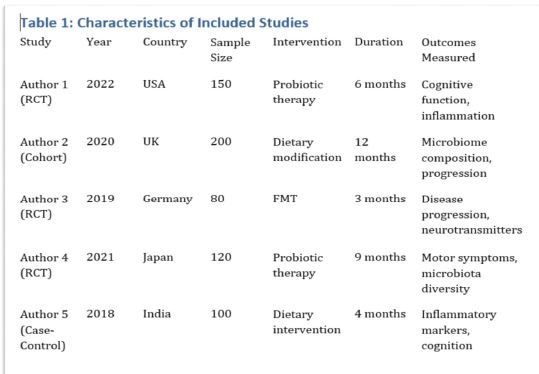
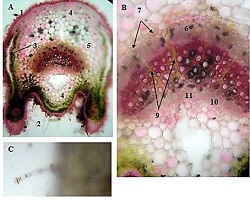
Rheumatoid arthritis (RA) is a multifactorial autoimmune disease of unknown etiology that mainly affects the joints, although extra-articular changes may occur. Pathogenic mechanism of development of rheumatoid arthritis has not been fully studied, which requires further research. Although the synovial membrane is a major target of arthritis, the synovial tissue-specific antigens that may elicit an immune response are virtually unknown. One of the most important reasons for studying the synovial membrane is to identify immunopathological processes that may be used as therapeutic targets and to predict outcomes. The pathological sign of rheumatoid arthritis is bone loss, which is manifested in the reduction of local, periarticular and systemic bone tissue and is the result of osteoclast induction and osteoblast suppression. Whether inflammation or an autoimmune mechanism is the main factor in bone damage is a controversial issue.
Downloads
A. Mathiessen and P. G. Conaghan, “Synovitis in osteoarthritis: current understanding with therapeutic implications.,” Arthritis Res Ther, vol. 19, no. 1, p. 18, Feb. 2017, doi: 10.1186/s13075-017-1229-9.
E. B. P. Lopes, A. Filiberti, S. A. Husain, and M. B. Humphrey, “Immune Contributions to Osteoarthritis.,” Curr Osteoporos Rep, vol. 15, no. 6, pp. 593–600, Dec. 2017, doi: 10.1007/s11914-017-0411-y.
E. Sanchez-Lopez, R. Coras, A. Torres, N. E. Lane, and M. Guma, “Synovial inflammation in osteoarthritis progression.,” Nat Rev Rheumatol, vol. 18, no. 5, pp. 258–275, May 2022, doi: 10.1038/s41584-022-00749-9.
Y.-J. Lin, M. Anzaghe, and S. Schülke, “Update on the Pathomechanism, Diagnosis, and Treatment Options for Rheumatoid Arthritis.,” Cells, vol. 9, no. 4, Apr. 2020, doi: 10.3390/cells9040880.
A. Najm et al., “Correction to: Standardisation of synovial biopsy analyses in rheumatic diseases: a consensus of the EULAR Synovitis and OMERACT Synovial Tissue Biopsy Groups.,” Arthritis Res Ther, vol. 20, no. 1, p. 282, Dec. 2018, doi: 10.1186/s13075-018-1795-5.
J. Hirose, H. Nishioka, M. Tsukano, S. Matsubara, K. Usuku, and H. Mizuta, “Matrix changes in articular cartilage in the knee of patients with rheumatoid arthritis after biological therapy: 1-year follow-up evaluation by T2 and T1ρ MRI quantification.,” Clin Radiol, vol. 73, no. 11, pp. 984.e11-984.e18, Nov. 2018, doi: 10.1016/j.crad.2018.06.020.
U. Harre and G. Schett, “[Association between bone catabolism and anti-citrullinated protein antibodies in rheumatoid arthritis].,” Z Rheumatol, vol. 75, no. 8, pp. 809–811, Oct. 2016, doi: 10.1007/s00393-016-0149-4.
J. C. Mora, R. Przkora, and Y. Cruz-Almeida, “Knee osteoarthritis: pathophysiology and current treatment modalities.,” J Pain Res, vol. 11, pp. 2189–2196, 2018, doi: 10.2147/JPR.S154002.
G. S. Dulay, C. Cooper, and E. M. Dennison, “Knee pain, knee injury, knee osteoarthritis & work.,” Best Pract Res Clin Rheumatol, vol. 29, no. 3, pp. 454–61, Jun. 2015, doi: 10.1016/j.berh.2015.05.005.
Q. Guo, Y. Wang, D. Xu, J. Nossent, N. J. Pavlos, and J. Xu, “Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies.,” Bone Res, vol. 6, p. 15, 2018, doi: 10.1038/s41413-018-0016-9.
T. Pap and A. Korb-Pap, “Cartilage damage in osteoarthritis and rheumatoid arthritis--two unequal siblings.,” Nat Rev Rheumatol, vol. 11, no. 10, pp. 606–15, Oct. 2015, doi: 10.1038/nrrheum.2015.95.
A. Sergijenko, A. J. Roelofs, A. H. K. Riemen, and C. De Bari, “Bone marrow contribution to synovial hyperplasia following joint surface injury.,” Arthritis Res Ther, vol. 18, p. 166, Jul. 2016, doi: 10.1186/s13075-016-1060-8.
M. D. Wechalekar and M. D. Smith, “Utility of arthroscopic guided synovial biopsy in understanding synovial tissue pathology in health and disease states.,” World J Orthop, vol. 5, no. 5, pp. 566–73, Nov. 2014, doi: 10.5312/wjo.v5.i5.566.
P. Luo et al., “The Role of Autophagy in Chondrocyte Metabolism and Osteoarthritis: A Comprehensive Research Review.,” Biomed Res Int, vol. 2019, p. 5171602, 2019, doi: 10.1155/2019/5171602.
O. Alvarez-Garcia, T. Matsuzaki, M. Olmer, L. Plate, J. W. Kelly, and M. K. Lotz, “Regulated in Development and DNA Damage Response 1 Deficiency Impairs Autophagy and Mitochondrial Biogenesis in Articular Cartilage and Increases the Severity of Experimental Osteoarthritis.,” Arthritis Rheumatol, vol. 69, no. 7, pp. 1418–1428, Jul. 2017, doi: 10.1002/art.40104.
W. Waldstein, G. Perino, S. L. Gilbert, S. A. Maher, R. Windhager, and F. Boettner, “OARSI osteoarthritis cartilage histopathology assessment system: A biomechanical evaluation in the human knee.,” J Orthop Res, vol. 34, no. 1, pp. 135–40, Jan. 2016, doi: 10.1002/jor.23010.
K. Okamoto et al., “Osteoimmunology: The Conceptual Framework Unifying the Immune and Skeletal Systems.,” Physiol Rev, vol. 97, no. 4, pp. 1295–1349, Oct. 2017, doi: 10.1152/physrev.00036.2016.
S. Garcia et al., “Colony-stimulating factor (CSF) 1 receptor blockade reduces inflammation in human and murine models of rheumatoid arthritis.,” Arthritis Res Ther, vol. 18, p. 75, Mar. 2016, doi: 10.1186/s13075-016-0973-6.
B. Wang et al., “IL-34 Upregulated Th17 Production through Increased IL-6 Expression by Rheumatoid Fibroblast-Like Synoviocytes,” Mediators Inflamm, vol. 2017, pp. 1–10, 2017, doi: 10.1155/2017/1567120.
S.-L. Wang, R. Zhang, K.-Z. Hu, M.-Q. Li, and Z.-C. Li, “Interleukin-34 Synovial Fluid Was Associated with Knee Osteoarthritis Severity: A Cross-Sectional Study in Knee Osteoarthritis Patients in Different Radiographic Stages.,” Dis Markers, vol. 2018, p. 2095480, 2018, doi: 10.1155/2018/2095480.
A. H. Hegaze, A. S. Hamdi, A. Alqrache, and M. Hegazy, “Efficacy of Platelet-Rich Plasma on Pain and Function in the Treatment of Knee Osteoarthritis: A Prospective Cohort Study.,” Cureus, vol. 13, no. 3, p. e13909, Mar. 2021, doi: 10.7759/cureus.13909.
Copyright (c) 2023 GEORGIAN SCIENTISTS

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.