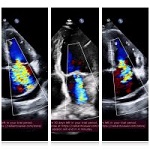
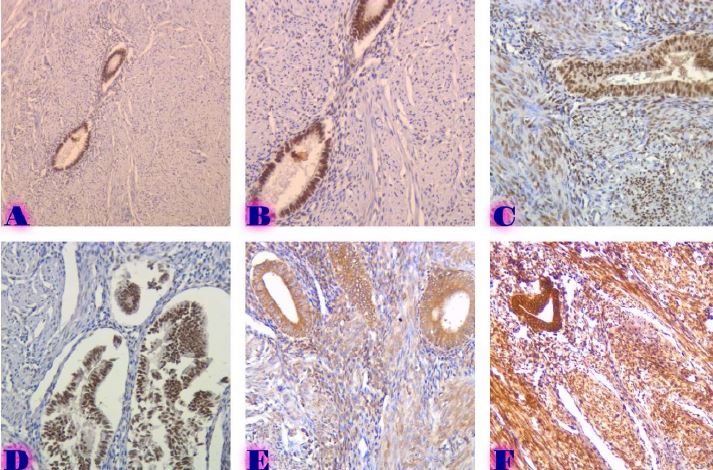
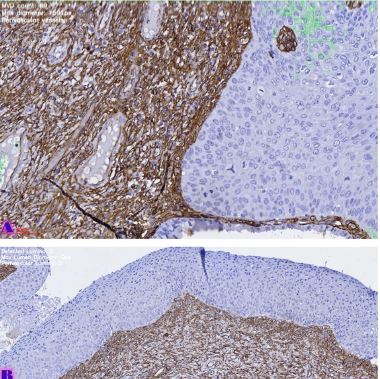
Left Ventricular Hypertrophy and Cardiomegaly in swimming exercised 10-11-week-old white experimental rats
Downloads
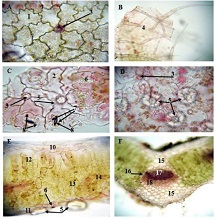
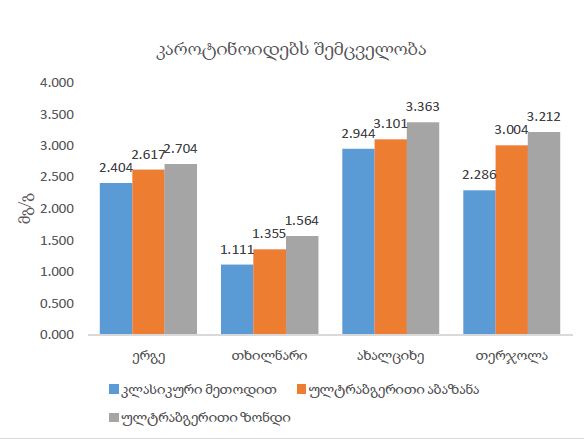
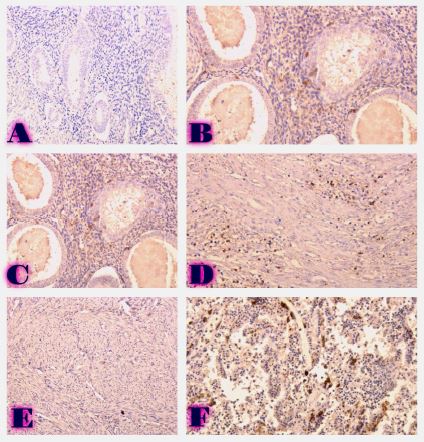
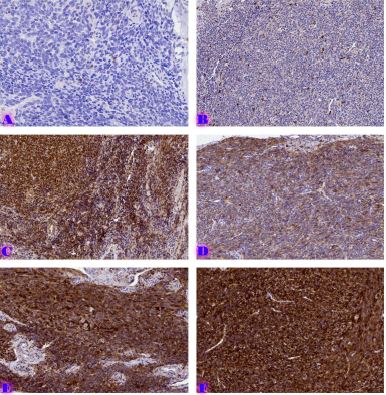
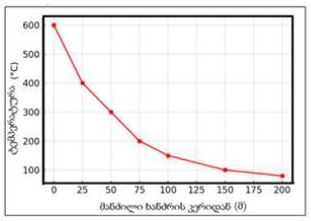
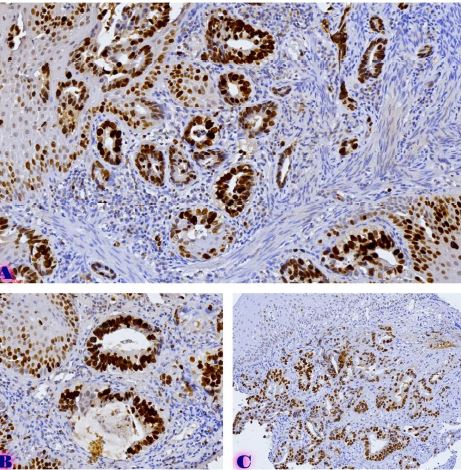
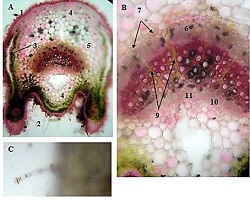
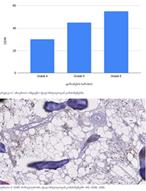
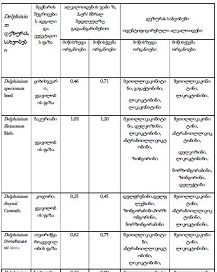
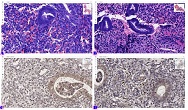
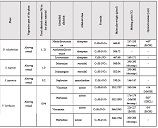
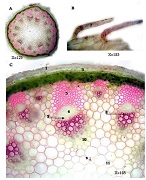
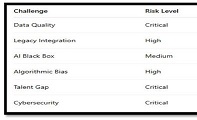
The aim of the study was to assess the quality of hyperthrophy of the left ventricle in 10-11 week-old rats after a 6-week dosed swimming load. In general, rat swimming is considered one of the best physiological triggers for hypertrophy of the left ventricle and end-diastolic volume increase. As part of our study, we ensured 10-11 week-old rats to undergo dosed swimming exercises for 6 weeks and afterward, we studied micro and macro-structural parameters of the heart in order to assess the quality of hypertrophy. The rats were divided into 3 groups: 1) a control group that did not receive any load (n=7); 2) The first study group was subjected to a daily 40-minute swimming load for 6 weeks with a weight in the amount of 2% of the body weight of the rat (n=7) attached to the tail, 3) The second study group, which was subjected to a daily 60-minute swimming exercise for 6 weeks, the weight of the load attached to the tail amounted to 4% of the weight of the rat (n=7). 3). Hypertrophy was observed only in rats of the experimental group (exercise load for 40 minutes with a load of 2% of body weight or exercise load for 60 minutes with a load of 4% of body weight). The ratio of heart weight to body weight is proportional to the intensity of load and to the increase in the duration of exercise. The increase in the mass of the heart was mainly due to an increase in the mass of the left ventricle. 60 minutes of histological exercise load induced hypertrophy in 7 of 7 rats. And a 40-minute load was sufficient for the development of left ventricular hypertrophy in 4 out of 7 rats included in the group. The mentioned study can be considered another step forward in the study of the pathogenetic mechanisms of myocardial hypertrophy and remodeling.
Downloads
Houser SR. When does spontaneous sarcoplasmic reticulum Ca2+ release cause a triggered arrhythmia? Cellular versus tissue requirements. Circ Res. 2000;87:725–7.
Mann DL, Bristow MR. Mechanisms and models in heart failure: The biomechanical model and beyond. Circulation. 2005;111:2837–49.
Pogwizd SM, Schlotthauer K, Li L, Yuan WL, Bers DM. Arrhythmogenesis and contractile dysfunction in heart failure - Roles of sodium-calcium exchange, inward rectifier potassium current, and residual beta-adrenergic responsiveness. Circ Res. 2001;88:1159–67.
Sipido KR, Volders PGA, Vos MA, Verdonck F. Altered Na/Ca exchange activity in cardiac hypertrophy and heart failure: a new target for therapy? Cardiovasc Res. 2002;53:782–805. https://animal.research.uiowa.edu/iacuc-guidelines-anesthesia
Bers DM. Cardiac excitation-contraction coupling. Nature. 2002;415:198–205.
Hilgemann DW. New insights into the molecular and cellular workings of the cardiac Na+/Ca2+ exchanger. Am J Physiol. 2004;287:C1167–C1172.
https://academic.oup.com/ilarjournal/article/62/1-2/238/6299201
Reuter H, Pott C, Goldhaber JI, Henderson SA, Philipson KD, Schwinger RHG. Na+-Ca2+exchange in the regulation of cardiac excitation-contraction coupling. Cardiovasc Res. 2005;67:198–207.
Gaughan JP, Furukawa S, Jeevanandam V, Hefner CA, Kubo H, Margulies KB, et al. Sodium/calcium exchange contributes to contraction and relaxation in failed human ventricular myocytes. Amer J Physiol. 1999;277:H714–H724.
Houser SR, Piacentino V, Weisser J. Abnormalities of calcium cycling in the hypertrophied and failing heart. J Mol Cell Cardiol. 2000;32:1595–607.
Terracciano CMN, DeSouza AI, Philipson KD, MacLeod KT. Na+ - Ca2+ exchange and sarcoplasmic reticular Ca2+ regulation in ventricular myocytes overexpressing the Na+ - Ca2+ exchanger. J Physiol London. 1998;512:651–67.
Weisser-Thomas J, Piacentino V, Gaughan JP, Margulies K, Houser SR. Calcium entry via Na/Ca exchange during the action potential directly contributes to contraction of failing human ventricular myocytes. Cardiovasc Res. 2003;57:974–85.
Hampton TG, Wang JF, DeAngelis J, Amende I, Philipson KD, Morgan JP. Enhanced gene expression of Na+/Ca2+ exchanger attenuates ischemic and hypoxic contractile dysfunction. Amer J Physiol. 2000;279:H2846–H2854.
Hobai IA, ORourke B. Enhanced Ca2+-activated Na+-Ca2+ exchange activity in canine pacing-induced heart failure. Circ Res. 2000;87:690–8.
Kent RL, Rozich JD, McCollam PL, McDermott DE, Thacker UF, Menick DR, et al. Rapid expression of the Na+-Ca2+ exchanger in response to cardiac pressure overload - rapid communication. Am J Physiol. 1993;265:H1024–H1029.
Schillinger W, Fiolet JW, Schlotthauer K, Hasenfuss G. Relevance of Na+-Ca2+ exchange in heart failure. Cardiovasc Res. 2003;57:921–33.
Studer R, Reinecke H, Bilger J, Eschenhagen T, Bohm M, Hasenfuss G, et al. Gene expression of the cardiac Na+ - Ca2+ exchanger in end-stage human heart failure. Circ Res. 1994;75:443–53.
Wang ZY, Nolan B, Kutschke W, Hill JA. Na+-Ca2+ exchanger remodeling in pressure overload cardiac hypertrophy. J Biol Chem. 2001;276:17706–11.
Zhang XQ, Song JL, Rothblum LI, Lun MY, Wang XJ, Ding F, et al. Overexpression of Na+/Ca2+ exchanger alters contractility and SR Ca2+ content in adult rat myocytes. Amer J Physiol. 2001;281:H2079–H2088.
Gomez AM, Valdivia HH, Cheng H, Lederer MR, Santana LF, Cannell MB, et al. Defective excitation-contraction coupling in experimental cardiac hypertrophy and heart failure. Science. 1997;276:800–6.
Schultz JEJ, Glascock BJ, Witt SA, Nieman ML, Nattamai KJ, Liu LH, Lorenz JN, et al. Accelerated onset of heart failure in mice during pressure overload with chronically decreased SERCA2 calcium pump activity. Am J Physiol. 2004;286:H1146–H1153.
Petit-Demouliere B, Chenu F, Bourin M. Forced swimming test in mice: a review of antidepressant activity. Psychopharmacology. (Berl) 2005;177:245–255.
David DJ, Renard CE, Jolliet P, Hascoet M, Bourin M. Antidepressant-like effects in various mice strains in the forced swimming test. Psychopharmacology (Berl) 2003;166:373–382.

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.