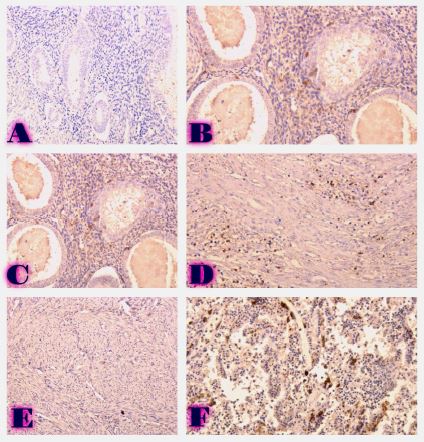
EFFECT OF REPETITIVE RFA ON THE IMMUNE RESPONSE OF THE PATIENTS WITH INOPERABLE PANCREATIC CANCER
Downloads
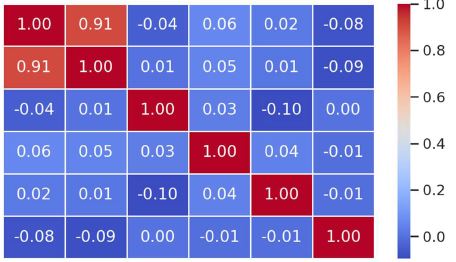
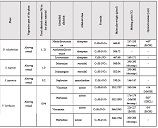
Annually, approximately 500,000 new cases of pancreatic ductal adenocarcinoma (PDAC) are diagnosed worldwide, with the same number of patients dying from this debilitating disease. Treatment resistance in PDAC results from the general immune suppression aggravated by the dense tumor stroma, which acts as a physical barrier to far too many treatment agents. Numerous studies suggest that RFA can trigger tumor-specific immune stimulation along with the disruption of the dense tumor stroma. Despite the immune stimulatory effect of RFA seen after procedure, a long-lasting clinical outcome of this procedure is hard to achieve. We aimed to explore the dynamics of immunological parameters in patients with PDAC treated with repetitive (three rounds of) RFA. Peripheral blood samples were analyzed from patients with inoperable pancreatic cancer who received three RFA treatments in a row, and compared to age-matched healthy controls. Only repetitive, and not a sole, administration of RFA actuated the decrease in protumorigenic cytokines (TGF-β, IL-17) and protumorigenic CD4+CD39+ and naive CD45RA T cells. Our study provides the first evidence on immunomodulatory benefits of repetitive RFA in patients with inoperable pancreatic cancer. Despite the fact that there was no other treatment used in the RFA group we still cannot attribute these immunologic changes to RFA alone. Larger scale studies involving different treatment modalities and complex immune readouts would guarantee more certainty to the clinical advantages of repetitive RFA.
Downloads
Rahib, L. et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 74, 2913–2921 (2014).
Siegel, R. L., Miller, K. D., Fuchs, H. E. & Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 71, 7–33 (2021).
Ryan, D. P. Treatment for potentially resectable exocrine pancreatic cancer. in UpToDate (eds. Goldberg, R. M. & Savarese, D. M. F.) (UpToDate, 2022).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 70, 7–30 (2020).
Wang, D. & DuBois, R. N. Immunosuppression associated with chronic inflammation in the tumor microenvironment. Carcinogenesis 36, 1085–1093 (2015).
Zhao, H. et al. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduction and Targeted Therapy 6, 1–46 (2021).
Ahmed, A. et al. Tertiary lymphoid structures and their association to immune phenotypes and circulatory IL2 levels in pancreatic ductal adenocarcinoma. Oncoimmunology 11, 2027148 (2022).
Zitvogel, L., Pietrocola, F. & Kroemer, G. Nutrition, inflammation and cancer. Nat. Immunol. 18, 843–850 (2017).
Shadhu, K. & Xi, C. Inflammation and pancreatic cancer: An updated review. Saudi J. Gastroenterol. 25, 3–13 (2019).
Toria, N., Kikodze, N., Rukhadze, R., Mizandari, M. & Chikovani, T. INFLAMMATORY BIOMARKERS IN PATIENTS WITH UNRESECTABLE PANCREATIC CANCER: A RETROSPECTIVE STUDY. Georgian Med. News 21–26 (2020).
Byrne, W. L., Mills, K. H. G., Lederer, J. A. & O’Sullivan, G. C. Targeting regulatory T cells in cancer. Cancer Res. 71, 6915–6920 (2011).
Zhang, H. et al. Paracrine SDF-1α signaling mediates the effects of PSCs on GEM chemoresistance through an IL-6 autocrine loop in pancreatic cancer cells. Oncotarget 6, 3085–3097 (2015).
Gabitass, R. F., Annels, N. E., Stocken, D. D., Pandha, H. A. & Middleton, G. W. Elevated myeloid-derived suppressor cells in pancreatic, esophageal and gastric cancer are an independent prognostic factor and are associated with significant elevation of the Th2 cytokine interleukin-13. Cancer Immunol. Immunother. 60, 1419–1430 (2011).
Hosein, A. N., Brekken, R. A. & Maitra, A. Pancreatic cancer stroma: an update on therapeutic targeting strategies. Nat. Rev. Gastroenterol. Hepatol. 17, 487–505 (2020).
Pook, H. & Pauklin, S. Mechanisms of Cancer Cell Death: Therapeutic Implications for Pancreatic Ductal Adenocarcinoma. Cancers 13, (2021).
Mizandari, M. et al. Interventional radiofrequency ablation: A promising therapeutic modality in the management of malignant biliary and pancreatic duct obstruction. J. Cancer 9, 629–637 (2018).
Bhardwaj, N. et al. Heat shock protein 70 expression following hepatic radiofrequency ablation is affected by adjacent vasculature. J. Surg. Res. 173, 249–257 (2012).
Geboers, B. et al. Needle-guided ablation of locally advanced pancreatic cancer: cytoreduction or immunomodulation by in vivo vaccination? Chin Clin Oncol 8, 61 (2019).
Janikashvili, N., Kikodze, N., Iobadze, M., Mazmishvili, K. & Chikovani, T. The Differential Effects of Radiofrequency Ablation and Hepatic Resection on Serum IL-10 Level in Patients with Liver Cancer. 03, (2019).
Levy, M. Y. et al. Cyclophosphamide unmasks an antimetastatic effect of local tumor cryoablation. J. Pharmacol. Exp. Ther. 330, 596–601 (2009).
Tempero, M. A. et al. Pancreatic adenocarcinoma, version 2.2014: featured updates to the NCCN guidelines. J. Natl. Compr. Canc. Netw. 12, 1083–1093 (2014).
Tempero, M. A. NCCN Guidelines Updates: Pancreatic Cancer. J. Natl. Compr. Canc. Netw. 17, 603–605 (2019).
David, C. J. & Massagué, J. Publisher Correction: Contextual determinants of TGFβ action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol. 19, 479 (2018).
Yan, X., Ten Dijke, P., Zhang, L. & Miyazawa, K. TGF-β and BMP Signaling in Cancer. (Frontiers Media SA, 2022).
Schulz, W. A. Molecular Biology of Human Cancers. (Springer Nature, 2023).
Grauel, A. L. et al. TGFβ-blockade uncovers stromal plasticity in tumors by revealing the existence of a subset of interferon-licensed fibroblasts. Nat. Commun. 11, 6315 (2020).
McAllister, F. et al. Oncogenic Kras activates a hematopoietic-to-epithelial IL-17 signaling axis in preinvasive pancreatic neoplasia. Cancer Cell 25, 621–637 (2014).
Hu, F. et al. IL-17 in pancreatic disease: pathogenesis and pharmacotherapy. Am. J. Cancer Res. 10, 3551–3564 (2020).
Karakhanova, S. et al. Characterization of myeloid leukocytes and soluble mediators in pancreatic cancer: importance of myeloid-derived suppressor cells. Oncoimmunology 4, e998519 (2015).
Loncle, C. et al. IL17 Functions through the Novel REG3β-JAK2-STAT3 Inflammatory Pathway to Promote the Transition from Chronic Pancreatitis to Pancreatic Cancer. Cancer Res. 75, 4852–4862 (2015).
Zhang, Y. et al. Immune Cell Production of Interleukin 17 Induces Stem Cell Features of Pancreatic Intraepithelial Neoplasia Cells. Gastroenterology vol. 155 210–223.e3 Preprint at https://doi.org/10.1053/j.gastro.2018.03.041 (2018).
Allard, D., Allard, B. & Stagg, J. On the mechanism of anti-CD39 immune checkpoint therapy. J Immunother Cancer 8, (2020).
Zhang, H. et al. The role of NK cells and CD39 in the immunological control of tumor metastases. Oncoimmunology 8, e1593809 (2019).
Canale, F. P. et al. CD39 Expression Defines Cell Exhaustion in Tumor-Infiltrating CD8 T Cells. Cancer Res. 78, 115–128 (2018).
Timperi, E. & Barnaba, V. CD39 Regulation and Functions in T Cells. Int. J. Mol. Sci. 22, (2021).
Hang, J. et al. The clinical implication of CD45RA+ naïve T cells and CD45RO+ memory T cells in advanced pancreatic cancer: a proxy for tumor biology and outcome prediction. Cancer Med. 8, 1326–1335 (2019).
Fernandes, A. T. G. et al. A prognostic value of CD45RA+, CD45RO+, CCL20+ and CCR6+ expressing cells as ‘immunoscore’ to predict cervical cancer induced by HPV. Sci. Rep. 11, 1–14 (2021).

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.