Biologic and Chemical Medicines – Differences in Terms of Basic Scientific and Regulatory Aspects
Downloads
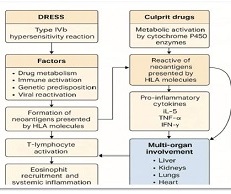
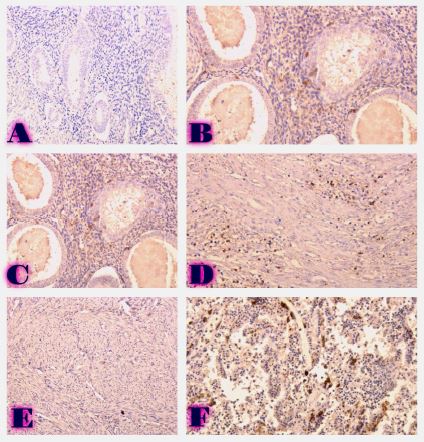
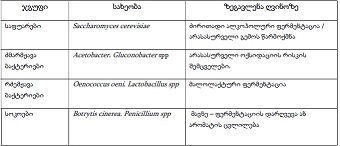
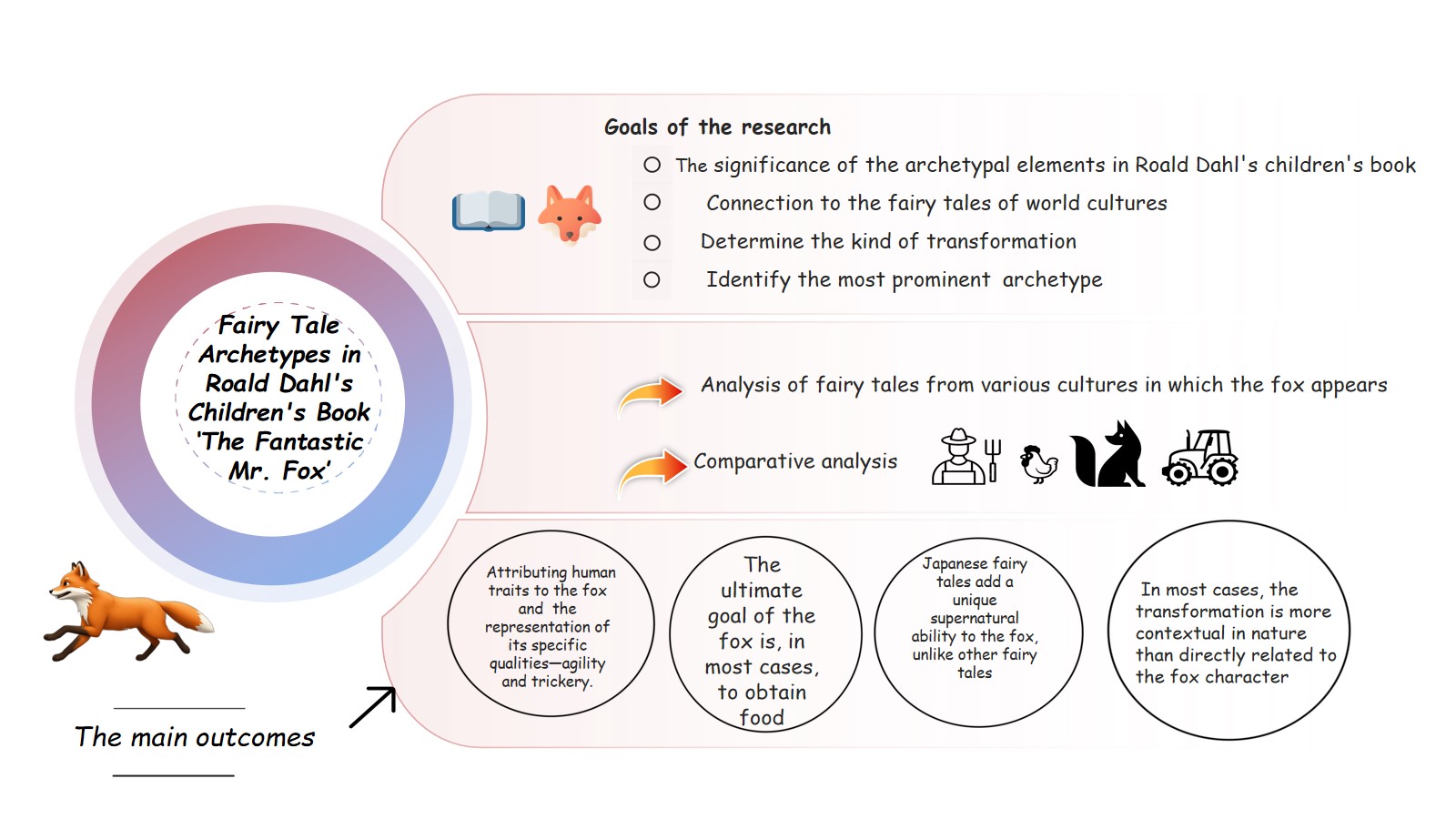
Biological Medicines represent an innovative advance in Healthcare, offering targeted and immunotherapies for a wide range of complex diseases. In this scientific article, we aim to present the fundamental differences between biological and chemical medicines in terms of mechanism of action, manufacturing process and regulatory environment. Additionally, we will discuss the basic scientific aspects of biosimilar and generic medicines, focusing on their regulatory details, which have a significant impact on patients care and healthcare system
Downloads
EMA. Biological Medicines https://www.ema.europa.eu/en/glossary/biological-medicine Accessed December. 2023
FDA. Biological Product Definitions
https://www.fda.gov/files/drugs/published/Biological-Product-Definitions.pdf Accessed December, 2023
Manufacturing Process of Biologics https://www.ema.europa.eu/en/documents/presentation/presentation-manufacturing-process-biologics-kowid-ho-afssaps_en.pdf Accessed December, 2023
Arnold G. Vulto, Orlando A. Jaquez - The process defines the product: what really matters in biosimilar design and production? Rheumatology 2017;56:iv14iv29
doi:10.1093/rheumatology/kex278
EMA. Generic and Hybrid Medicines
https://www.ema.europa.eu/en/human-regulatory-overview/marketing-authorisation/generic-and-hybrid-medicines. Accessed December, 2023
FDA. Generic Drugs
https://www.fda.gov/drugs/buying-using-medicine-safely/generic-drugs. Accessed December, 2023
FDA. Biosimilars.
https://www.fda.gov/drugs/therapeutic-biologics-applications-bla/biosimilars. Accessed December, 2023
Guidelines on evaluation of similar biotherapeutic products (SBPs). In: WHO Expert Committee on Biological Standardization: Sixtieth report. Geneva: World Health Organization; 2013: Annex 2 (WHO Technical Report Series, No. 977;
http://www.who.int/biologicals/publications/trs/areas/biological_therapeutics/TRS_977_Annex_2.pdf, accessed December 2023).
EMA. Biosimilars overview. https://www.ema.europa.eu/en/human-regulatory-overview/biosimilar-medicines-overview. Accessed December 2023
EMA. Similar biological medicinal products- Scientific guideline https://www.ema.europa.eu/en/similar-biological-medicinal-products-scientific-guideline. Accessed December, 2023
EMA. Biosimilars in EU. https://www.ema.europa.eu/en/documents/leaflet/biosimilars-eu-information-guide-healthcare-professionals_en.pdf Accessed December, 2023
ICH Q5E Biotechnological/biological products subject to changes in their manufacturing process: comparability of biotechnological/biological products – Scientific guidelinehttps://www.ema.europa.eu/en/ich-q5e-biotechnological-biological-products-subject-changes-their-manufacturing-process-comparability-biotechnological-biological-products-scientific-guideline. Accessed December, 2023
International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH Harmonised Tripartite Guideline. Specifications: test procedures and acceptance criteria for biotechnological/biological products Q6B. 1999 guideline recommended for adoption https://www.ema.europa.eu/en/ich-q6b-specifications-test-procedures-and-acceptance-criteria-biotechnological-biological-products-scientific-guideline. Accessed December 2023
EMA. Immunogenicity assessment of biotechnology-derived therapeutic proteins – Scientific guideline. https://www.ema.europa.eu/en/immunogenicity-assessment-biotechnology-derived-therapeutic-proteins-scientific-guideline. Accessed December, 2023
WHO. Monoclonal Antibodies (mABs) https://www.who.int/teams/health-product-policy-and-standards/standards-and-specifications/monoclonal-antibodies. Accessed December, 2023
Copyright (c) 2024 Georgian Scientists

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.