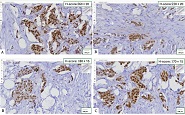
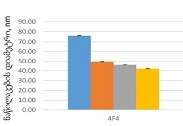
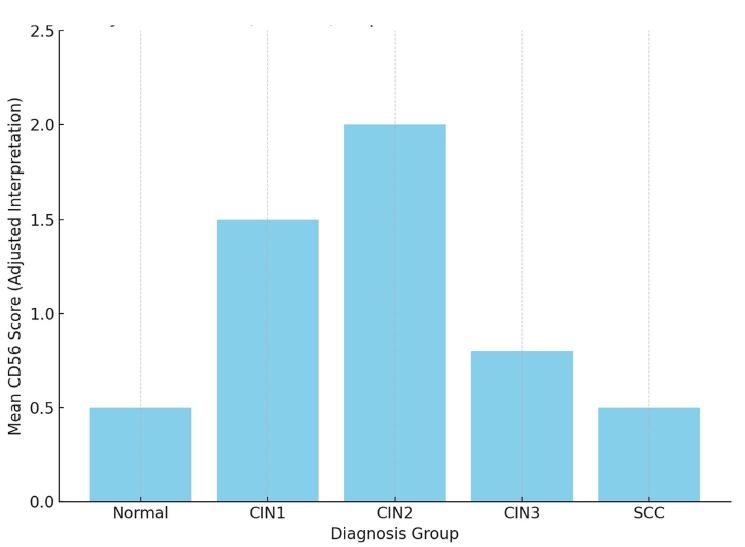
Cross-senolytic effects of dasatinib and quercetin in humans
Downloads
Additional Files
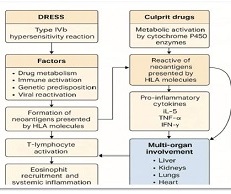
Introduction: Senescent cells are toxic cells. Their number increases every day and after some time it leads to the appearance of signs of aging. Senescent cells cause age-related pathologies.
Objective: Simultaneous administration of dasatinib and quercetin in Ercc1-/∆ mice resulted in the partial killing of senolytic cells and improved and prolonged health, age-related symptoms and pathologies. We decided to test the senolytic effect of the combination of dasatinib and quercetin in humans.
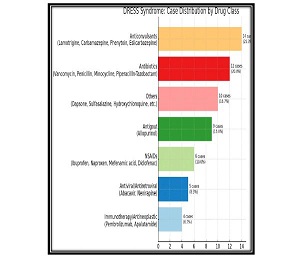
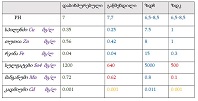
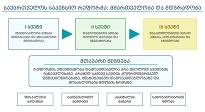
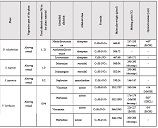
Study Design: A clinical study was conducted on 64 male volunteers aged 36 years and older. Our volunteers were divided into 4 groups, each with 16 people. The D+Q group received 50 mg dasatinib and 500 mg quercetin together once daily for 5 days. Other groups: D+P - dasatinib + placebo group; Q+P – quercetin + placebo group; P+P – placebo (dasatinib) + placebo (quercetin) group.
Methods: The study was completed by screening a blood count and a stair ascending test to register and evaluate the changes.
Results: Co-administration of 50 mg dasatinib with 500 mg quercetin showed a clear senolytic effect. This was confirmed by the improved results of the stair ascending test. A general blood test showed that the combination of these two drugs in such a dosage is most likely to be harmless.
Conclusion: These results demonstrate the possibility of selective elimination of senescent cells in humans and the effectiveness of senolytic agents in improving health and physical performance.
Downloads
Abumuslimov et al., (1994). An electron microscopic study of centriole and centrosome morphogenesis in the early development of the mouse. Tsitologiia.
Bruning, (2013). Inhibition of mTOR signaling by quercetin in cancer treatment and prevention. Anticancer Agents Med Chem.
Campisi and d’Adda di Fagagna, (2007). Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol.
Chichinadze, K. N., & Tkemaladze, D. V. (2008). Centrosomal hypothesis of cellular aging and differentiation. Advances in Gerontology= Uspekhi Gerontologii, 21(3), 367-371. PMID: 19432168
Chichinadze, K., Tkemaladze, D., & Lazarashvili, A. (2012a). New class of RNA and centrosomal hypothesis of cell aging. Advances in Gerontology= Uspekhi Gerontologii, 25(1), 23-28. PMID: 22708440
Chichinadze, K., Tkemaladze, J., & Lazarashvili, A. (2012b). Discovery of centrosomal RNA and centrosomal hypothesis of cellular ageing and differentiation. Nucleosides, Nucleotides and Nucleic Acids, 31(3), 172-183. doi: 10.1080/15257770.2011.648362. PMID: 22356233
Chichinadze, K., Tkemaladze, J., & Lazarashvili, A. (2012c). A new class of RNAs and the centrosomal hypothesis of cell aging. Advances in Gerontology, 2(4), 287-291
Chichinadze, K., Lazarashvili, A., & Tkemaladze, J. (2013). RNA in centrosomes: structure and possible functions. Protoplasma, 250(1), 397-405. doi: 10.1007/s00709-012-0422-6. Epub 2012 Jun 10. PMID: 22684578
Coppé et al., 2008. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. DOI: 10.1371/journal.pbio.0060301
Coppé et al. 2010. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. DOI: 10.1146/annurev-pathol-121808-102144.
Hartung and Stahl, 1977. Preleptotene chromosome condensation in mouse oogenesis. Cytogenet Cell Genet. DOI: 10.1159/000130777
Hayflick L, 1997. Mortality and immortality at the cellular level. A review. Biochemistry (Mosc).
Ishiuchi and Torres-Padilla, 2013. Towards an understanding of the regulatory mechanisms of totipotency. Curr Opin Genet Dev. DOI: 10.1016/j.gde.2013.06.006.
Jaba, T. (2022). Dasatinib and quercetin: short-term simultaneous administration yields senolytic effect in humans. Issues and Developments in Medicine and Medical Research Vol. 2, 22-31. doi: https://doi.org/10.9734/bpi/idmmr/v2/15155D
Justice et all., 2019. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine. DOI: 10.1016/j.ebiom.2018.12.052.
Lezhava, T., Monaselidze, J., Jokhadze, T., Kakauridze, N., Khodeli, N., Rogava, M., Tkemaladze J., ... & Gaiozishvili, M. (2011). Gerontology research in Georgia. Biogerontology, 12, 87-91. doi: 10.1007/s10522-010-9283-6. Epub 2010 May 18. PMID: 20480236; PMCID: PMC3063552
Matsaberidze, M., Prangishvili, A., Gasitashvili, Z., Chichinadze, K., & Tkemaladze, J. (2017). To topology of anti-terrorist and anti-criminal technology for educational
Montero et al., 2011. Inhibition of SRC family kinases and receptor tyrosine kinases by dasatinib: possible combinations in solid tumors. Clin Cancer Res. DOI: 10.1158/1078-0432.CCR-10-2616.
Olave et al., Upstream stimulatory factor-2 mediates quercetin-induced suppression of PAI-1 gene expression in human endothelial cells. J Cell Biochem. DOI: 10.1002/jcb.22760.
Prangishvili, A., Gasitashvili, Z., Matsaberidze, M., Chkhartishvili, L., Chichinadze, K., Tkemaladze, J., ... & Azmaiparashvili, Z. (2019). System components of health and innovation for the organization of nano-biomedic ecosystem technological platform. Current Politics and Economics of Russia, Eastern and Central Europe, 34(2/3), 299-305.
Roos et al., 2016. Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell. DOI: 10.1111/acel.12458.
Roy and Kundu, 2014. Gene regulatory networks and epigenetic modifications in cell differentiation. IUBMB Life. doi: 10.1002/iub.1249
Teh and Aziz, 2000. A stair-climb test of cardiorespiratory fitness for Singapore. Singapore Med J. PMID: 11296784
Tkemaladze, J., & Chichinadze, K. (2005). Potential role of centrioles in determining the morphogenetic status of animal somatic cells. Cell biology international, 29(5), 370-374. doi: 10.1016/j.cellbi.2005.03.003. PMID: 15886028.
Tkemaladze, J. V., & Chichinadze, K. N. (2005). Centriolar mechanisms of differentiation and replicative aging of higher animal cells. Biochemistry (Moscow), 70, 1288-1303. doi: 10.1007/s10541-005-0261-6. PMID: 16336191
Tkemaladze, J., & Chichinadze, K. (2010). Centriole, differentiation, and senescence. Rejuvenation research, 13(2-3), 339-342. doi: 10.1089/rej.2009.0904. PMID: 20426623
Tkemaladze, J., Tavartkiladze, A., & Chichinadze, K. (2012). Programming and Implementation of Age-Related Changes. In Senescence. IntechOpen. DOI: 10.5772/33420
Tkemaladze, J., & Apkhazava, D. (2019). Dasatinib and quercetin: short-term simultaneous administration improves physical capacity in human. J Biomedical Sci, 8(3), 3.
Tkemaladze, J. (2022). Long-Term Differences between Regenerations of Head and Tail Fragments in Schmidtea Mediterranea Ciw4. Available at SSRN 4257823.
Tkemaladze, J. (2023). Reduction, proliferation, and differentiation defects of stem cells over time: a consequence of selective accumulation of old centrioles in the stem cells?. Molecular Biology Reports, 50(3), 2751-2761.doi: 10.1007/s11033-022-08203-5. Epub 2022 Dec 30. PMID: 36583780
Tkemaladze, J. (2023). The centriolar hypothesis of differentiation and replicative senescence. Junior Researchers, 1(1). doi: https://doi.org/10.52340/2023.01.01.15
Tkemaladze, J. (2023). Structure and possible functions of centriolar RNA with reference to the centriolar hypothesis of differentiation and replicative senescence. Junior Researchers, 1(1), 156–170. https://doi.org/10.52340/2023.01.01.17
Zhu et al., 2015. The Achilles' heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. DOI: 10.1111/acel.12344.
Прангишвили, А. И., Гаситашвили, З. А., Мацаберидзе, М. И., Чичинадзе, К. Н., Ткемаладзе, Д. В., & Азмайпарашвили, З. А. (2017). К топологии антитеррористических и антикриминальных технологии для образовательных программ. In Управление развитием крупномасштабных систем MLSD'2017 (pp. 284-287).
Прангишвили, А. И., Гаситашвили, З. А., Мацаберидзе, М. И., Чхартишвили, Л. С., Чичинадзе, К. Н., Ткемаладзе, Д. В., ... & Азмайпарашвили, З. А. (2017). Системные составляющие здравоохранения и инноваций для организации европейской нано-биомедицинской екосистемной технологической платформы. In Управление развитием крупномасштабных систем MLSD'2017 (pp. 365-368).
Ткемаладзе Д. , Цомаиа Г ., Жоржолиани И. (2001). Создание искусственных самоадаптирующихся систем на основе Теории Прогноза. Искусственный интеллект. УДК 004.89. Искусственный интеллект. УДК 004.89. https://www.ipai.net.ua/uk/arch-2001-3#
Ткемаладзе, Д. В., & Чичинадзе, К. Н. (2005). Центриолярные механизмы дифференцировки и репликативного старения клеток высших животных. Биохимия, 70(11), 1566-1584.
Чичинадзе, К., Ткемаладзе, Д., & Лазарашвили, А. (2012). Новый класс рнк и центросомная гипотеза старения клеток. Успехи геронтологии, 25(1), 23-28.
Чичинадзе, К. Н., & Ткемаладзе, Д. В. (2008). Центросомная гипотеза клеточного старения и дифференциации. Успехи геронтологии, 21(3), 367-371
Copyright (c) 2023 GEORGIAN SCIENTISTS

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.