WAYS OF LEARNING OF IDEAL STATE EQUATION (METHODOLOGICAL MATERIAL)
Downloads
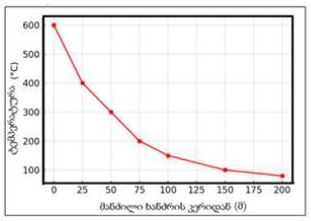
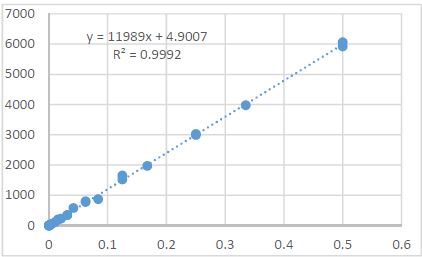
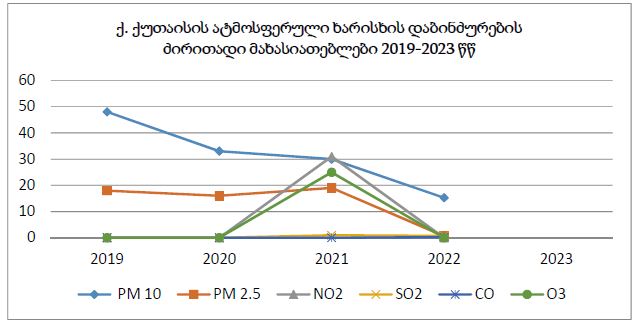
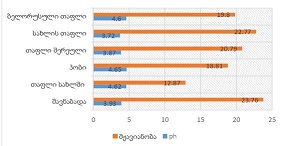
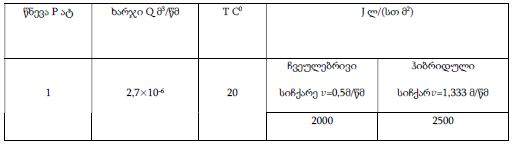
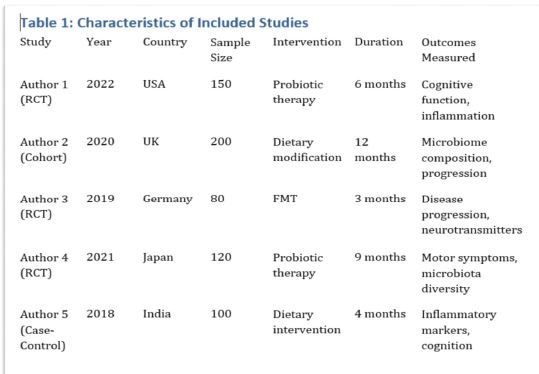
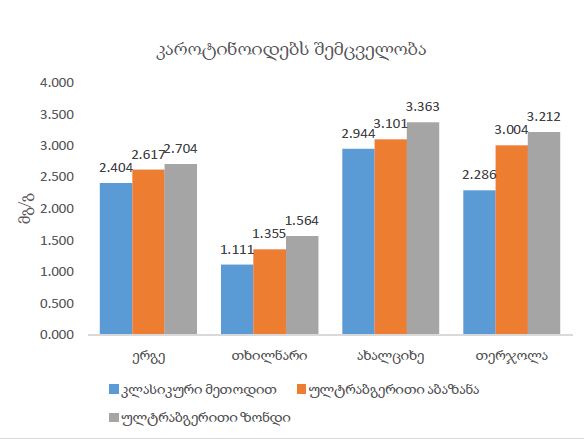
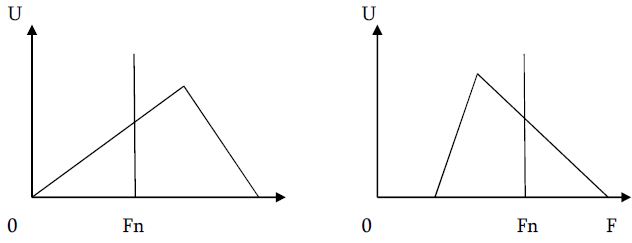
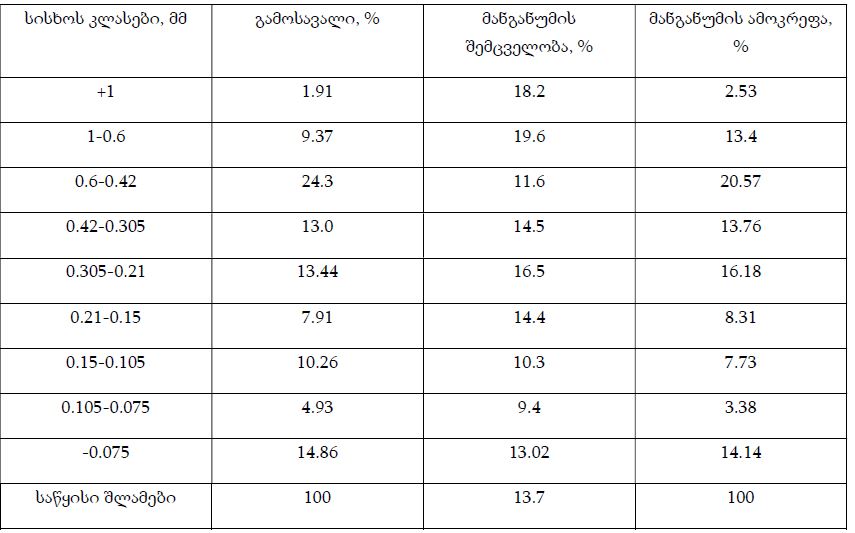
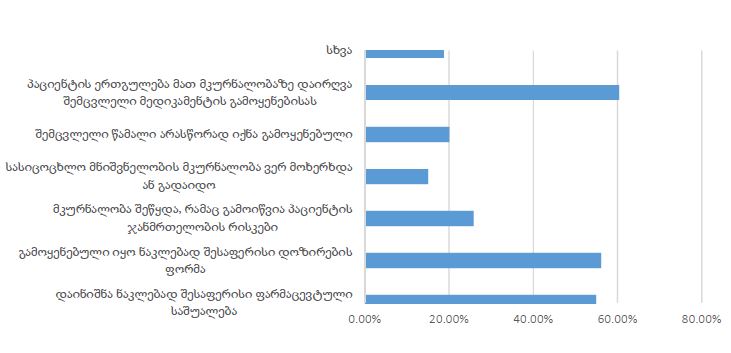
The article presents methods of obtaining the Mendeleev-Clapeyron equation. Thermodynamics considers the macrosystem as a whole and describes its properties not on the basis of the microstructure of the substance, but on the basis of the experiment establishes a connection between the macroscopic parameters of the substance. This method is distinguished by its simplicity, but it has a drawback, which is that its internal mechanism is not explained in the thermodynamic discussion of events. For example, one can experimentally obtain a relationship between the pressure and volume of an ideal gas at constant temperature pV=const, but why this is so can only be explained by the methods of molecular physics.
Downloads
https://courses.lumenlearning.com/suny-physics/chapter/13-2-thermal-expansion-of-solids-and-liquids/
ქ. ტატიშვილი. ფიზიკა. მე-11 კლასი. მოსწავლის წიგნი. გამომცემლობა დიოგენე. 2012, 191 გვ.
თ. ბჟალავა, თ. ფაღავა, მ. მეცხვარიშვილი. ზოგადი ფიზიკის მოკლე კურსი. საგამომცემლო სახლი „ტექნიკური უნივერსიტეტი“. 2016, 142 გვ.
ა. გიგინეიშვილი, გ. კუკულაძე. ზოგადი ფიზიკა. I ტომი. საგამომცემლო სახლი „ტექნიკური უნივერსიტეტი“. 2011, 372 გვ.
Copyright (c) 2023 GEORGIAN SCIENTISTS

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.