FORMULATION AND TECHNOLOGY OF ENTERIC AND FOAM-FORMING RABEPRAZOLE TABLETS
Downloads
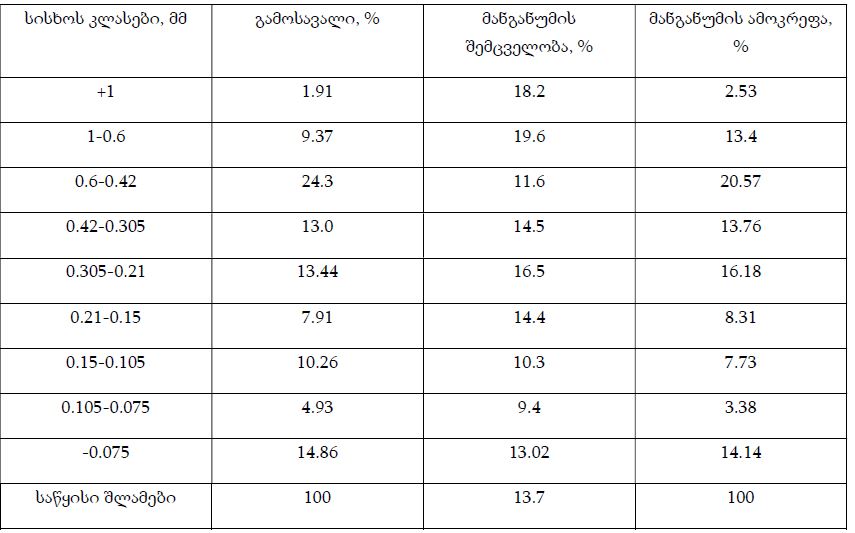
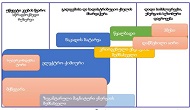
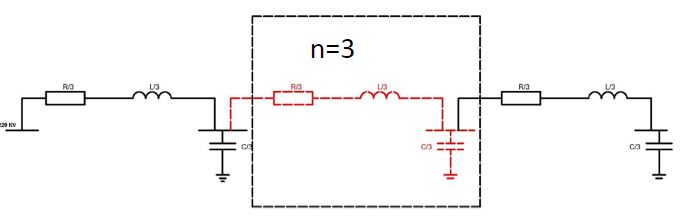
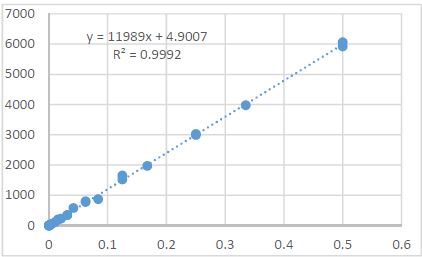
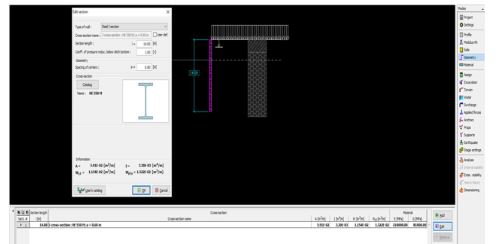
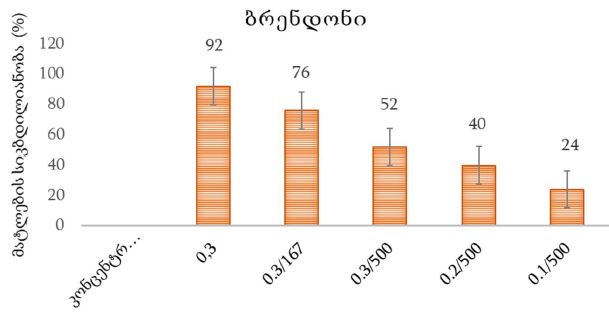
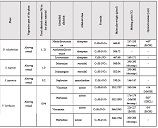
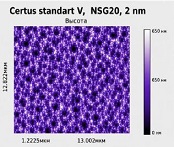
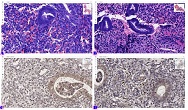
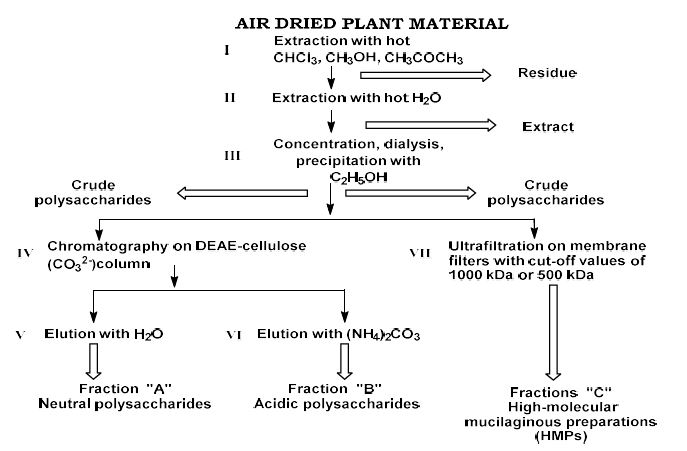
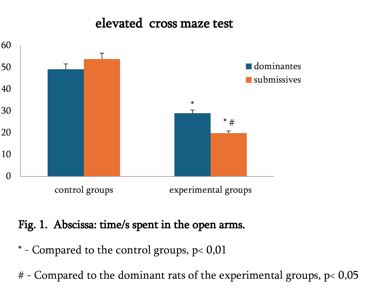
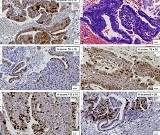
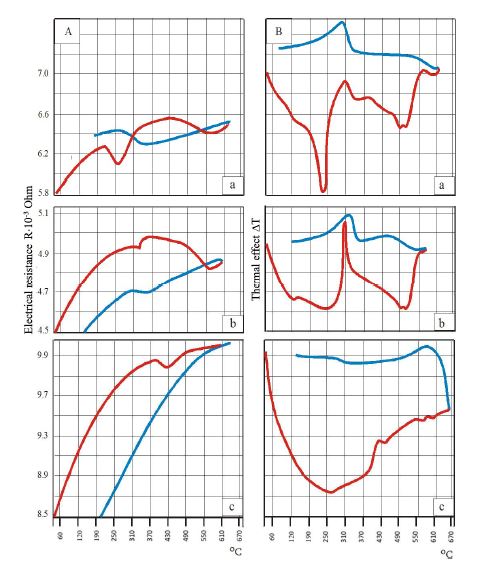
Helicobacter pylori (H. pylori) is considered the main cause of gastroduodenal diseases. For the eradication of Helicobacter, triple therapy is actively used, the so-called "Gold standard", which combines antibiotics and proton pump inhibitors. Among the innovative delivery systems for the gastroenterological drug formulationsfoams appear to be of particular interest.Foams are light systems. Unlike solid medicationforms, theyare not simply depositedin the gastrointestinal mucosa, on the contrary, they grow in volume,have a large contact surface, completely cover the mucous membrane, and penetrate it. Targeted delivery, enteric-soluble and foam-forming tablets have been developed from the "Gold standard"ingredients with a significant amount of antacids. The study aimed to determine the composition of enteric-coated and foam-forming rabeprazole sodium tablets and process their technology. Based on biopharmaceutical research, the optimal composition of foam-forming powder has beenformulated as an auxiliary substance for the preparation of foam-forming tablets, with the following composition: sodium lauryl sulfate, lecithin, and METHOCEL™ E6 PLV (0.75:1.25:5) 7%, Xanthan, and guar gum in a ratio of 1:1 - 2%, Isomalt 10%. Based on biopharmaceutical studies, the formulations of enteric-soluble and foam-forming core,tabletcoating composition, and manufacturing technologyof rabeprazolehave beendrawn up. The physical, chemical, and technological properties of foam-forming and enteric-coated rabeprazole tablets have been studied. And, we have ascertainedthat the technology appliedfor their manufacturing meets the quality requirements of this medication: its appearance, dosing accuracy, mechanical strength, solubility, foaming, stability, etc. The dissolution profile of rabeprazole tablets, generated at pH 1.2 and 6.8,has been studied. We have ascertainedthat Active Pharmaceutical Ingredient (API)is almost 100% resistant to the acidic environment: the amount of API released during 2 hours in the acid area is insignificant and varies within 3%. The API of the tablet is completely released within 30 min at pH 6.
Downloads
Assaad, S., Chaaban, R., Tannous, F. et al. Dietary habits and Helicobacter pylori infection: a cross sectional study at a Lebanese hospital. BMC Gastroenterol 18, 48 (2018). https://doi.org/10.1186/s12876-018-0775-1
Zhu Y, Zhou X, Wu J, Su J, Zhang G. Risk Factors and Prevalence of Helicobacter pylori Infection in Persistent High Incidence Area of Gastric Carcinoma in Yangzhong City. Gastroenterol Res Pract. 2014;2014:481365. doi: 10.1155/2014/481365.
Suzuki H, Nishizawa T, Hibi T. Helicobacter pylori eradication therapy. Future Microbiol. 2010 Apr; 5(4):639-48. doi: 10.2217/fmb.10.25. PMID: 20353303.
Tsay FW, Hsu PI. H. pylori infection and extra-gastroduodenal diseases. J Biomed Sci. 2018 Aug 29;25(1):65.
Jorge Morcillo Muñoz, William Otero Regino, Martín Gómez Zuleta, How can Helicobacter pylori eradication therapies be improved? Rev Col Gastroenterol vol. 33 n o.4 Bogotá Oct./ Dec. 2018
Enzo Lerardi, Giuseppe Losurdo, Rosa Federica La Fortezza, Mariabeatrice Principi, Michele Barone, Alfredo Di Leo //Optimizing proton pump inhibitors in Helicobacter pylori treatment: Old and new tricks to improve effectiveness. World J Gastroenterol. 2019 Sep 14;25(34):5097-5104.
Yi Hu, Yin Zhu* and Nong-Hua Lu*// Novel and Effective Therapeutic Regimens for Helicobacter pylori in an Era of Increasing Antibiotic Resistance, Front. Cell. Infect. Microbiol., 05 May 2017.
Verma A, Dubey J, Hegde RR, Rastogi V, Pandit JK. Helicobacter pylori: past, current and future treatment strategies with gastroretentive drug delivery systems. J Drug Target. 2016 Dec; 24(10):897-915. doi: 10.3109/1061186X.2016.1171326.
Veerapandian Chandrasekar, John Swamy Gabriela, K. Kannan and A. Sangamithra. Effect of foaming agent concentration and drying temperature on physiochemical and antimicrobial properties of foam mat dried powder. Asian J. Dairy & Food Res., 34 (1) 2015: 39-43
Zazadze R., Bakuridze L., Chavelashvili L., Gongadze N., Bakuridze A. DEVELOPMENT OF FORMULATION AND TECHNOLOGY OF FOAMING AGENT FROM MASTIC (PISTACIA LENTISCUS L.) GUM. Georgian Medical News. 2022.
REVAZ ZAZADZE, LASHA BAKURIDZE, DALI BERASHVILI, ALIOSHA BAKURIDZE. DETERMINAT-ION OF STOM¬ACH SOLUBLE AND FOAMING RABEPRAZOLE AND METRONIDAZOLE TABLETS FOR-MULATION. Experimental & Clinical Medicine. 2022.
რევაზ ზაზაძე. „Helicobacter Pylori-ის ერადიკაციის „ოქროს სტანდარტისა“ და დანამასტაკის გუმფისისაგან მიზანმიმართული მიწოდების და ქაფწარმომქმნელი ტაბლეტების შემუშავება“. დისერტაცია ფარმაციის დოქტორის აკადემიური ხარისხის მოსაპოვებლად. თბილისი 2022 წელი 194 გვ.
Copyright (c) 2023 GEORGIAN SCIENTISTS

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.